2674
Views & Citations1674
Likes & Shares
In plants, it is well established that chloroplast
is one of the main organelles to break down during leaves senescing by drought.
The effects of exogenous salicylic acid and/or trehalose on flag leaf growth,
total chlorophylls, lipid peroxidation and thylakoid membranes in flag leaves
were investigated in drought-stressed wheat cultivars; Gemmieza-7 (drought
sensitive cultivar) and Sahel-1 (drought tolerant cultivar) plants. Water
stress induced a significant reduction in growth vigor of flag leaf, especially
its biomass, the degree of succulence as well as leaf area and specific leaf
area, total chlorophylls, lipid peroxidation and chloroplasts number. On the
other hand, drought resulted in a marked increase in oleosomes volume in
mesophyll cells of flag leaves in both wheat cultivars when compared with those
of the well-watered plants. Gemmieza-7 appeared to be the most affected
cultivars. Application of SA and/or Tre appeared to act in a reverse manner by
inducing an additional increase in the values of abovementioned criteria.
Furthermore, electron microscopic examination of mesophyll cells of wheat flag
leaf revealed that water stress negatively affected the chloroplasts and
oleosomes ultrastructure in the two cultivars during grain-filling. The
chloroplasts showed to be more or less spherical with irregular shape after
drought stress treatment. Moreover, disorganized membrane system with swollen
thylakoids was identified. These changes in membrane structure are mainly due to
the rapid oxidative damage evaluated as malondialdehyde. Moreover, many
plastoglobuli were found in the chloroplasts of droughted plants than those
recognized in control plants. This effect was more conspicuous with the
sensitive one. The exogenous application of SA and/or Tre had a positive effect
on the ultrastructure characteristics of mesophyll cells of wheat flag leaves.
Keywords: Wheat;
Drought; Oleosomes; Chloroplasts ultrastructure
Abbreviations: SA:
Salicylic Acid; Tre: Trehalose; TAGs: Triacylglycerols
INTRODUCTION
Flag leaf plays an important role in wheat
plant life as it exports assimilates to spike and developing grains [1].
Reduction in the rate of leaf surface expansion, followed by a cessation of
expansion is the general pattern of plant response to stress [2].
Permanent membrane integrity and function under a given level of
dehydration has been used as a measure of drought tolerance [3]. Loss of water,
degradation of photosynthetic pigment and lipid peroxidation are also
significantly stimulated due to drought stress [4].
So far, most studies on these cultivars have been concerned with their
morphological characteristics but few ultrastructural studies have addressed
their tolerance to drought stress. Therefore, in the light of the
above-mentioned survey and keeping in view the importance of wheat and water
stress, it was of particular interest to assess up to what extent salicylic
acid and/or trehalose could ameliorate the deleterious effects of drought
stress on both wheat plants. Furthermore, the main goal of this study is to
find out the positive effect of grain priming with salicylic acid and spraying
the wheat plants with trehalose on increasing the drought tolerance of
sensitive cultivar Gemmieza-7.
MATERIALS AND
METHODS
Plant material and
growth conditions
Two wheat genotype, Gemmieza-7 (drought sensitive cultivar) and Sahel-1
(drought tolerant cultivar) were selected. The sterilized grains from each
cultivar were divided into two sets (≈ 500 g per set for each cultivar). Grains
of the 1st set were soaked in distilled water to serve as a control, while
those of the 2nd were soaked in salicylic acid (3 mM) for about 6 h.
Grains were raised in plastic pots (20 cm in diameter) filled with 5.5 kg soil
(clay/sand 2/1, v/v), where grains were sown (20 November 2011 and 2012) in
each pot. The pots were then kept in a greenhouse at research area of Botany
Department, Faculty of Science. The plants were subjected to natural day/night
conditions (minimum/maximum air temperature and relative humidity were 15/25°C and 35/45%, respectively) at
mid-day during the experimental period. The plants were irrigated to field
capacity with tap water.
On the day 65 after planting (at the beginning of heading) the pots of
the 1st set was allocated to four groups (20 pots per each group) as
follows: control (cont.), water stress (WS), trehalose control, trehalose+water
stress (trehalose+WS). The 2nd set group was allocated to four
groups as follows: salicylic acid control (SA), salicylic acid+water stress
(SA+WS), control trehalose+salicylic acid (SA+trehalose) and salicylic
acid+trehalose+water stress (SA+trehalose+WS). For trehalose (1.5 mM)
treatment, the plants were sprayed by trehalose 48 h before starting the stress
period and weekly during the stress period.
Agronomic traits were calculated using the following formula:
Leaf area = Length × Breadth × 0.75 [7],
Specific leaf area = Leaf area / Dry mass [8],
Degree of succulence = Water amount / Leaf area [9],
Degree of sclerophylly = Dry mass / Leaf area [10]
Measuring the plant photosynthetic pigment (chl a+chl b) was according
to the method of Arnon [11]. Estimation of lipid peroxidation was assayed
spectrophotometrically using thiobarbituric acid-malondialdehyde assay
(TBA-MDA) [12].
Ultrastructural
Studies Using Transmission Electron Microscope
The leaf tissues were processed for transmission electron microscopy
TEM according to the method of Woods and and Gay [13]. The plant tissues were cut
into 1 mm2 pieces and fixed in 2.5% glutaraldehyde in 0.1 M sodium
cacodylate buffer for 24 h at room temperature, and then post-fixed in 1%
osmium tetroxide in the same buffer. Leaf tissues were dehydrated in a graded
ethanol series and infiltrated with Araldite. Sections showed silver-grey
interference colours cut by Reichert ultramicrotome were collected on copper
grids, stained with uranyl acetate and lead citrate [14]. Sections were
examined and photographed by using Jeol 1010 TEM.
Counting of Chloroplasts
Number
MEASUREMENT OF
OLEOSOMES VOLUME IN SEMI-THIN SECTIONS
A new technique developed using the image analysis for measuring the
volume of oleosomes (in semi-thin sections from flag leaf mesophyll tissues was
estimated to a leaf or cell unit volume for each treatment) was performed.
STATISTICAL ANALYSIS
The data were subjected to one-way analysis of variance (ANOVA), and
different letters indicate significant differences between treatments at p ≤
0.05, according to CoHort/CoStat software, Version 6.311.
RESULTS
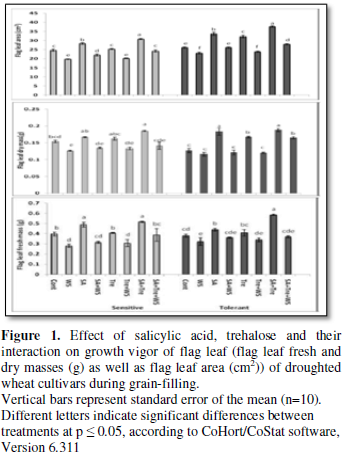
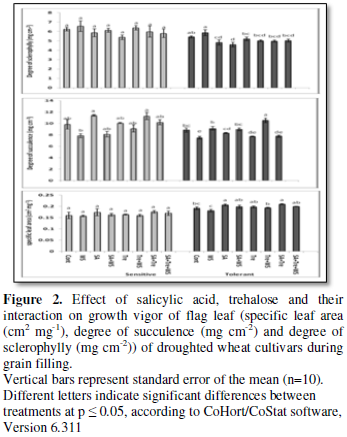
Changes in growth
vigor of flag leaf
Perusal of the data shown in Figures
1 and 2 cleared that, in comparison with control values, water stress
resulted in general significant reduction (p ≤ 0.05) in growth vigor of flag
leaf, especially its biomass (fresh and dry masses), degree of succulence as
well as leaf area and specific leaf area (non-significant increase in case of
sensitive cultivar) of both wheat cultivars during grain-filling. On the other
hand, water stress induced a clear increase (p ≤ 0.05) in the degree of leaf
sclerophylly of tolerant cultivar and a non-significant increase in case of the
sensitive cultivar. Application of SA and/or Tre resulted in the enhancement of
flag leaf growth (i.e., biomass, area and specific area as well as the degree
of succulence) in well watered and water stressed wheat plants. Also, the application
of these chemicals to the water-stressed plants caused a noticeable decrease (p
≤ 0.05) in the degree of flag leaf sclerophylly intolerant cultivar and a
non-significant decrease in case of sensitive cultivar beyond that of either
control or stressed plants. Generally, SA and Tre treatment appeared to be the
most effective treatment in counteracting the negative effects of water stress
on all flag leaf growth criteria.
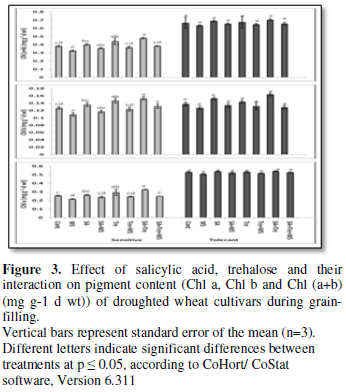
Changes in pigment
content
Changes in lipid peroxidation
Changes in
chloroplasts number in mesophyll cells of wheat flag leaf
Changes in the
ultrastructure of chloroplasts and oleosomes of flag leaf of wheat cultivars
sensitive wheat cultivar
Transmission electron microscopic examination of well-watered wheat
flag leaf revealed that the shape of chloroplast from mesophyll cells of
control plants was comparatively regular (tended to be oval or elliptical to
somewhat) when compared with that of droughted wheat plants. Chloroplast
contained starch grains and plastoglobuli. More specifically, mitochondrion was
also observed. Grain presoaking in SA caused the chloroplast of well-watered
wheat plants to be more regular. Chloroplast contained starch grains and a
continuous "end-to-end" distribution of chloroplasts around the cell
periphery was observed. More specifically, chloroplasts also contained
plastoglobuli. Conspicuous spherical globules, the oleosomes, appeared to be
free in the vacuole of mesophyll cells. These inclusions had a sharply-defined
osmiophilic interface and apparently lack a limiting membrane.
Regarding the ultrastructure of the flag leaf of well-watered wheat
plants treated with Tre, the chloroplasts were normal and ellipsoidal in shape.
Chloroplast was closely associated with the cell wall and the membrane system
of grana and intergranal lamellae was somewhat organized with defined
chloroplast envelope. Starch grains had approximately as the same size as those
of control plants, but more in its number. Conspicuous spherical oleosomes
appeared to be free in the vacuole of mesophyll cells. Application of an
interaction of SA and Tre to well-watered plants resulted in regular or oval
chloroplasts containing starch grains. A continuous "end-to-end" distribution
of chloroplasts around the cell periphery was observed. Furthermore, the
chloroplast contained plastoglobuli and appeared to be dividing. More
mitochondria were also observed.
Regarding the ultrastructure of the flag leaf of stressed wheat plants
treated with Tre, the chloroplast shape was, like in SA treatment, more regular
than the stressed ones. Chloroplasts were closely associated with the cell wall
and the membrane system was slightly affected. They contained plastoglobuli
which are increased in number and become bigger. Oleosomes were more spherical
in shape and their interface was getting thicker and appeared more osmiophilic.
More mitochondria were also identified. Application of an interaction of SA and
Tre to water-stressed plants resulted in very slight changes in chloroplasts.
They kept their ellipsoidal shape as the control. They were still closely
associated with the cell wall with few exceptions and showed the organized
membrane system. Chloroplasts contained bigger plastoglobuli.
Tolerant wheat
cultivar:
Regarding control tolerant cultivar, the chloroplasts were normal and ellipsoidal
in shape. Although the membrane system of grana and intergranal lamellae was
not clear enough, they also contained bigger plastoglobuli and starch grains. A
continuous "end-to-end" distribution of chloroplasts around the cell
periphery was observed. Conspicuous oleosomes appeared to be free in the
vacuole of mesophyll cells and contained electron-dense material. These
inclusions had a sharply-defined osmiophilic interface and apparently lacked a
limiting membrane. Grain presoaking in SA caused the chloroplast of
well-watered wheat plants to be more regular. Although the membrane system of
grana and intergranal lamellae was not clear enough, chloroplasts contained
plastoglobuli and starch grains. Mitochondria were also identified. Conspicuous
oleosomes appeared to be free in the vacuole of mesophyll cells and contained
electron-dense material. These inclusions had a sharply-defined osmiophilic
interface and apparently lacked a limiting membrane.
Regarding the ultrastructure of the flag leaf of stressed wheat plants
treated with Tre, the chloroplast shape was, like in SA treatment, more regular
than the stressed ones. They kept their ellipsoidal shape as the control. They
were still closely associated with the cell wall with few exceptions and showed
the organized membrane system. They also contained many starch grains and few
plastoglobuli. Small spherical oleosomes apparently lack a limiting membrane
was found. Oleosomes appeared to be more ellipsoidal in shape and no changes in
their ultrastructure were pronounced. Many mitochondria were also observed.
Application of an interaction of SA and Tre to water-stressed plants resulted
in very slight changes in chloroplasts. They kept their ellipsoidal shape as
the control. Chloroplast contained starch grains and a continuous
"end-to-end" distribution of chloroplasts around the cell periphery
was observed and they were still closely associated with the cell wall. They
also contained many and bigger plastoglobuli. Conspicuous spherical oleosomes
had a sharply-defined osmiophilic interface and apparently lacks a limiting
membrane was observed.
DISCUSSION
Data clearly showed that drought-induced a drastic reduction in Chl a+b
of the sensitive wheat cultivar and a non- significant decrease in drought
tolerant cultivar wheat plants. In accordance with these results, Aldesuquy et
al. [18] reported that when the leaves of wheat plants started to senesce,
there was a gradual decline in chlorophylls [18]. Moreover, Netondo et al. [16]
explained that the decline in leaf chlorophylls induced by water stress may
occur through the decrease of its synthesis and/or the increase of its degradation.
Salicylic acid and/or Tre treatments enhanced chl a chl b, and Chl a+b,
production in stressed or unstressed wheat plants. In general, the interaction
of SA and Tre appeared to be the most effective treatment in enhancing the
pigment production in flag leaf of stressed and unstressed wheat plants.
Similar results were observed by ShiraniBidabadi et al. [19]. The manipulating
effect of SA and Tre may be due to the fact that SA increases leaf longevity of
droughted plants by keeping their chlorophylls content, therefore delay their
senescence. In relation to these results, Chandra and Bhatt [20] showed that
the increasing or decreasing effect of SA on chlorophyll content of cowpea.
Moreover, Alam et al. [21] reported that interaction of Tre with drought
improved chl (a+b) contents in Brassica species.
Drought stress caused a significant increase in lipid peroxidation.
This increase lipid peroxidation my result from malondialdehyde (MDA) increased
as those obtained by Aldesuquy and Ghanem [22] and Fazeli et al. [23]. On
contrary, Salicylic acid and/or Tre treatments mitigated the ill effect of
drought on lipid membrane degradation by reducing malondialdehyde (MDA).
Oleosomes, globules rich in neutral lipids, were present in flag leaf
of untreated and treated wheat plants where there was a noticeable increase in
oleosomes volume in control and droughted plants in both cultivars. Drought led
to massive increase in oleosomes volume in mesophyll cells of flag leaf of both
cultivars during grain-filling. The pattern of increase was higher in
Gemmieza-7 than Sahel-1. This increase in oleosomes accompanied drought stress
could be attributed to the fact that water stress often induces premature
senescence and subsequently decreasing the storing fats through increases its
hydrolysis by lipase into fatty acids and glycerol.
It is evident from the current studies that a number of chloroplasts
were adversely influenced by drought in both wheat cultivars with more
reduction in chloroplasts number in mesophyll cells of flag leaf in both
cultivars when compared with those of the control plants. The most pronounced
effect was recorded for the sensitive cultivar. On the other hand, application
of SA and/or Tre caused an additional increase in these values. The most
pronounced effect was recorded for the interaction of SA and Tre treatment.
This observation was in accordance with the results obtained by Smethurst et
al. [24]. This reduction may be a strategy of protection and/or acclimation.
Moreover, this reduction may be a consequence of the reduction in the leaf area
of wheat plants under salt stress.
Transmission electron microscopic examination of wheat flag leaf
revealed that the shape of chloroplasts in well-watered plants was comparatively
regular with a well-organized membrane system. On the other hand, the
chloroplasts in the leaves of droughted plants began to be less or more
spherical with an irregular shape. Water-stressed cells also displayed
extensive but thin peripheral cytoplasmic regions devoid of chloroplasts. While
a continuous distribution of chloroplasts around the cell's periphery was
observed in control plants, the chloroplasts of droughted ones appeared
discrete. Clear matching with those obtained by Stoyanova et al. [25] who
studied leaf ultrastructure in maize plants grown under water stress.
Furthermore, similar observations under different stress forms were also
reported by Sam et al. [26] and Smethurst et al. [24].
With the lower availability of CO2 to chloroplasts, ROS build up and
in-turn can increase lipid peroxidation of the chloroplast, inducing damage to
thylakoid membranes and pigment breakdown resulting in the reduction of
photosynthesis [25].
Stressing wheat plants with drought resulted in the appearance of
plastoglobuli within the chloroplasts. During senescence, the number of
plastoglobules increased as the thylakoids break down. Plastoglobule numbers
also increased in plants subjected to environmental conditions [27,28].
Hormones involved directly in the control of chloroplast biogenesis and
function and affect chloroplast ultrastructure, pigment production and finally
the rate of carbon assimilation [30].
CONCLUSION
1. Aldesuquy HS, Baka ZA, Mickky BM
(2014) Kinetin and spermine mediated induction of salt tolerance in wheat
plants: Leaf area, photosynthesis and chloroplast ultrastructure of flag leaf
at ear emergence. Egypt J Basic Appl Sci 1: 77-87.
2. Parida AK, Das AB (2005) Salt
tolerance and salinity effects on plants: Review. Ecotoxicol Environ Saf 60:
324-349.
3. Shinde BM, Limaye AS, Deore GB,
Laware SL (2010) Physiological responses of groundnut (Arachis hypogaea L.)
varieties to drought stress. Asian J Exp Biol 1: 65-68.
4. Kadioglu A, Saruhan N, Sağlam A,
Terzi R, Acet T (2011) Exogenous salicylic acid alleviates effects of long term
drought stress and delays leaf rolling by inducing antioxidant system. Plant
Growth Regul 64: 27-37.
5. Buchholz A (2006) Characterization
of the diffusion of non-electrolytes across plant cuticles: Properties of the
lipophilic pathway. J Exp Bot 57: 2501-2513.
6. Munne-Bosch S, Alegre L (2004) Die
and let live: Leaf senescence contributes to plant survival under drought
stress. Functional Plant Biol 31: 203-216.
7. Quarrie SA, Jones HG (1979)
Genotype variation in leaf water potential, stomatal conductance and abscisic
acid concentration in spring wheat subjected to artificial drought stress. Ann
Bot 44: 323-332.
8. Beadle CL (1993) Growth analysis.
In: Photosynthesis and production in a changing environment. A field and
laboratory manual (Hall DC, Scurlock JMO, Bolhar HR, Leegod RC and Long SP
eds.). Chapman and Hall, London, pp. 36-46.
9. Delf EM (1912) Transpiration in
succulent plants. Ann Bot 26: 409-440.
10. Witkoswski ETF, Lamont BB (1991)
Leaf specific mass confounds leaf density and thickness. Oecologia 84: 362-370.
11. Arnon DI (1949) Copper enzymes in
isolated chloroplasts: Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:
1-15.
12. Hodges DM, DeLong JM, Forney C,
Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay
for estimating lipid peroxidation in plant tissues containing anthocyanin and
other interfering compounds. Plantae 207: 604-611.
13. Woods AM, Gay JL (1983) Evidence
for a neckband delimiting structural and physiological regions of the host
plasma membrane associated with haustoria of Albugo candida. Physiol Mol Plant
Pathol 23: 73-88.
14. Juniper BE, Cox GC, Gilchrist AJ,
Williams PK (1970) Techniques for plant electron microscopy. Blackwell
Scientific Publications, Oxford.
15. Shani U, Ben-Gal A (2005)
Long-term response of grapevines to salinity: Osmotic effects and ion toxicity.
Am J Enol Viticult 56: 148-154.
16. Netondo GW, Onyango JC, Beck E
(2004) Sorghum and salinity. II. Gas exchange and chlorophyll fluorescence of
sorghum under salt stress. Crop Sci 44: 806- 811.
17. Welch ME, Rieseberg LH (2002)
Habitat divergence between a homoploid hybrid sunflower species, Helianthus
paradoxus (Asteraceae) and its progenitors. Am J Bot 89: 472-479.
18. Aldesuquy HS, Abo-Hamed SA, Abass
MA, Elhakem AH (2012) Role of glycine betaine and salicylic acid in improving
growth vigour and physiological aspects of droughted wheat cultivars. J Stress
Physiol Biochem 8: 150-171.
19. ShiraniBidabadi S, Mahmood M,
Baninasab B, Ghobadi C (2012) Influence of salicylic acid on morphological and
physiological responses of banana (Musa acuminata cv. Berangan, AAA) shoot tips
to in vitro water stress induced by polyethylene glycol. Plant Omics J 5:
33-39.
20. Chandra A, Bhatt RK (1998)
Biochemical and physiological response to salicylic acid in relation to the
systemic acquired resistance. Photosynthetica 45: 255-258.
21. Alam MM, Nahar K, Hasanuzzaman M,
Fujita M (2014) Trehalose-induced drought stress tolerance: A comparative study
among different Brassica species. Plant Omics J 7: 271-283.
22. Aldesuquy HS, Ghanem HE (2015)
Exogenous alicylic acid and trehalose ameliorate short term drought stress in
wheat cultivars by up-regulating membrane characteristics and antioxidant
defense system. J Horticult 2: 2.
23. Fazeli F, Ghorbanli M, Niknam V
(2007) Effect of drought on biomass, protein content, lipid peroxidation and
antioxidant enzymes in two sesame cultivars. Biologia Plantarum 51: 98-103.
24. Smethurst CF, Gill WM, Shabala S
(2009) Using excised leaves to screen lucerne for salt tolerance: Physiological
and cytological evidence. Plant Signal Behav 4: 39-41.
25. Stoyanova D, Elena TE, Ivan YI
(2002) Influence of different soil moisture on anatomy of maize leaves and
ultrastructure of chloroplasts. Bulgarian J Plant Physiol 28: 11-20.
26. Sam O, Ramirez C, Coronado MJ,
Testillano PS, Risueno MC (2003) Changes in tomato leaves induced by NaCl
stress: Leaf organization and cell ultrastructure. Biologia Plantarum 47:
361-366.
27. Mostowska A (1997) Environmental
factors affecting chloroplasts. In: Handbook of photosynthesis (Pessarakli M,
ed.). Dekker, New York, USA, pp: 407-426.
28. Austin JR, Frosta E, Vidic PA,
Kesslerc F, Staehelina LA (2006) Plastoglobules are lipoprotein subcompartments
of the chloroplast that are permanently coupled to thylakoid membranes and
contain biosynthetic enzymes. Plant Cell 18: 1693-1703.
29. Kang G, Wang Z, Xia K, Sun G
(2007) Protection of ultrastructure in chilling-stressed banana leaves by
salicylic acid. Journal of Zhejiang University Science B 8: 277-282.
30.
Yaronskaya
E, Vershilovskaya I, Poers Y, Alawady AE, Averina N, et al. (2006) Cytokinin
effects on tetrapyrrole biosynthesis and photosynthetic activity in barley
seedlings. Planta 224: 700-709.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Genetics and Cell Biology (ISSN:2639-3360)
- Journal of Astronomy and Space Research
- Food and Nutrition-Current Research (ISSN:2638-1095)
- Journal of Womens Health and Safety Research (ISSN:2577-1388)
- Journal of Veterinary and Marine Sciences (ISSN: 2689-7830)
- Journal of Microbiology and Microbial Infections (ISSN: 2689-7660)
- Journal of Biochemistry and Molecular Medicine (ISSN:2641-6948)