2644
Views & Citations1644
Likes & Shares
Plant
pathogens and insect herbivores attaching and surviving on plant and their
produce can associate indirectly with each other and interact directly or
indirectly resulting in serious economic loss to the growers. In this
experiment the influence of co-interaction between systemic plant pathogen Botrytis cinerea Pears Fr (Helotiales
Sclerotiniaceae) and herbivorous insect green peach aphid Myzus persicae on the growth of lettuce Lactuca sativa (Asteraceae: Compositae) was investigated. Studies
showed that the co- interaction between Botrytis
cinerea and Myzus persicae causes
stress on the lettuce plants resulting in economic loss. The studies found that
co-interaction between B. cinerea and
M. persicae resulted in lower lesion
of B. cinerea and M. persicae counts when both were
present on same host plants. In addition the stress resulting from co-
interaction affected the growth of lettuce plant resulting in a significant
reduction in plant height, internode length, leaf size and also significant
reduction of fresh shoot and root weight.
Keywords: Botrytis cinerea, Co-interaction, Plant growth, Myzus persicae
INTRODUCTION
Myzus persicae and Botrytis cinerea an herbivorous insect and plant pathogenic fungus
respectively are of great economic importance due to their ability to spread diseases
causing loss of plants and its produce [1-3]. Therefore, the ability of Botrytis cinerea and Myzus persicae to survive on the same
plant host makes them to exhibit a co-interaction.
Myzus persicae Sulzer (Hemiptera: Aphididae), the
common green peach potato aphid, is found throughout the world, where it is
principally regarded as
a serious pest of many important agricultural crops because of its
ability to transmit plant viruses [4]. It feeds on crops such as peaches, potatoes, sugar beet, tobacco and various ornamental
crops grown in landscapes and in glasshouses and most lettuce
plants [4,5]. A high
population of M. persicae (Figure 1) on a crop causes injury by
removing large volumes of sap from the plants and depleting them of needed
nutrients [4]. In addition, they also cause indirect injury by the production
of sugary honeydew which makes the leaves susceptible to microbial attack,
which then reduces leaf quality [6].
Botrytis cinerea is a systemic plant pathogen with a wide
host range of more than 500 plants [7]. It is the causal agent of grey-mold
disease and causes greater economic losses, during both pre and post-harvest
phase of plant growth, more than any other disease [2,8], it is also
responsible for about 20% losses of affected crops worldwide [2]. It is
reported to cause annual crop losses worldwide ranging from $10-$100 billion
[7]. Plant and their produce stored for weeks or months at temperatures ranging
from 0-10°C and transported fruit, small fruits, ornamental flowers and bulbs,
as well as forest seedlings are all attacked and destroyed by B. cinerea [9-11]. Therefore, Williamson
et al. [12] concluded that B. cinerea
as the most widely distributed disease of vegetables, ornamental fruits and
field crops throughout the world. The pathogen produces clear or grey conidia
on branched conidiophores which are dispersed by humid air currents, splashing
water, tools and clothing; the conidiophores initiate a new infection on
healthy plants [2,13]. The conidia of the fungus can infect the seedlings,
flowers, stems or leaves through wounded or senescing tissues and directly
through the epidermis [2,14]. The symptoms of the pathogen may sometimes appear
very
The lettuce Lactuca sativa L. (Asteraceae (Compositae)) is a significant
horticultural crop, widely used as source of food [18-20]. Many researchers
have reported studies of Botrytis cinerea
on crops such as strawberries, kiwifruits, raspberries, grapes and others [15].
Lettuces seem to have received little attention with respect to their infection
by B. cinerea and yet they share the
devastating effect caused by B. cinerea
under favorable conditions, especially in protected crops. The aphids affecting
lettuce crops are a problem worldwide causing serious economic losses [21,22].
Higher number of aphids may result in stunted plant growth, but, the most
damaging effect of aphid attack is wilting and head contamination, which reduce
the market value of the lettuce [21]. Despite the fact that lettuce is attack
by several species of aphids however, the most common aphid pest of lettuce is
the green peach-potato aphid (Myzus persicae Sulzer) [4,21].
The co-interactional interaction of insect herbivores
and pathogenic fungi inhabiting the same host plant occur in a situation where
the first attacker alters the fitness of the plant host in a way that it
affects the second attacker, while in return the second attacker induces the
plant host to synthesised chemicals which reduces the growth of the first
attacker all at the expense of other important plant processes [23-26].
However, the success of these types of interaction is dependent on the type and
behavior of insect herbivore and fungal pathogen involved [27,28]. Therefore two hypotheses were
tested which are: (i) Decrease in the spread and expression of B. cinerea and lower growth rate of
aphids will occur following stress on the host lettuce plant by the
co-interaction of systemic pathogen B. cinerea
and aphids M. persicae. (ii)
Reduction of plant height and dry mass of the plant will occur as a result of
stress on the plant host due to co-interaction of systemic plant pathogens and
aphids.
MATERIALS AND
METHODS
Study plants
Sterilized seed of lettuce was sown in 80, 15 cm diameter pots filled
with a vermiculite growing medium in a controlled environment room (18-20°C,
ambient humidity and 12-14 h L: D). Half of the plants (40, plants) were grown
from clean uninfected seeds while the remaining half were grown from seed
systemically infected with B. cinerea.
Plant infestation
with the Myzus persicae
One month after germination all the
experimental plants were infested with ten nymphs of aphids Myzus persicae Sulzer (Hemiptera
Aphididae). The M. persicae were reared for three generation for proper
effect of telescoping of generation before being used in the experiment [29].
Infestation was done by placing the aphids on the reverse side of the leaves
(20 infected and 20 uninfected plants) using moist brush. In order to prevent
the escape of the aphids immediately after infestation plants were covered with
a vented plastic container. The remaining uninfested plants (20 infected and 20
uninfected) served as controls.
Population size of Myzus persicae
The population size of M. persicae was obtained by counting the number of aphids on the
plants. The counting was done once a week for twenty weeks, starting four weeks
after infestation. Also visual examination was used to assess the appearance of
B. cinerea infection on the plants.
Plant height
Plant height was taken from all 80 plants in
the four treatments. Height of the plants was measured before harvest using
measuring tape.
Leaf size
Leaf size was taken from all 80 plants in the
four treatments. Before harvest leaf size was measured from the two fully
expanded middle leaves using measuring tape.
Internode length
Before harvest Internode length was taken
from all the 80 plants in the four treatments. Before harvest length of the
internode was measured from all the plants by using measuring tape.
Dry shoot weight (g)
Harvested shoots were removed from all the
plants and washed under running tap water and allowed to dry in an oven for one
hour. Thereafter, dry shoot weight was taken from all the plants using an
electronic balance (Kern scale Technic, 440-21N).
Dry root weight (g)
The roots were washed under running tap water
and allowed to dry in an oven for 1 h. The measurements were taken using an
electronic balance (Kern scale Technic, 440-21N).
STATISTICAL ANALYSIS
All data were analysed using ANOVA with
post-hoc Tukey tests [30]. As the data from plant height, internode length,
leaf size, fresh shoot and root weight did not meet assumptions of normality, a
Box-Cox approach was used to determine the correct transformation prior to
analysis. The data from plant height and internode length were log transformed
while data from leaf size and fresh shoot weight was square root transformed
before the analysis. Root weight was Ln transformed. Number of aphid colony
survivorship between infected and uninfected plants was analysed using two ways
ANOVA [31].
RESULTS
Expression of
systemic B. cinerea lesion on M. persicae infested and uninfected
plants
More lesion of B. cinerea was recorded on plants uninfected by aphids than on
infested plants. However, uninfected plants were free of lesions. Therefore,
the presence of B. cinerea
significantly reduces the plant height, leaf size, internode length and plant
dry weight (Table 1).
Number of Myzus persicae on infected and
uninfected plants by systemic B. cinerea
Size of plant parts
Influence of
systemic B. cinerea and M. persicae on plant height: There was a significant
effect of aphid infestation status and B.
cinerea infection status on the height of the plant was obtained from the
experimental plants in which the presence of either resulted in a significant
reduction in plant height. The interaction term was not significant (Figure 1).
Knowledge of
students about lawns in the campus environment
Influence of
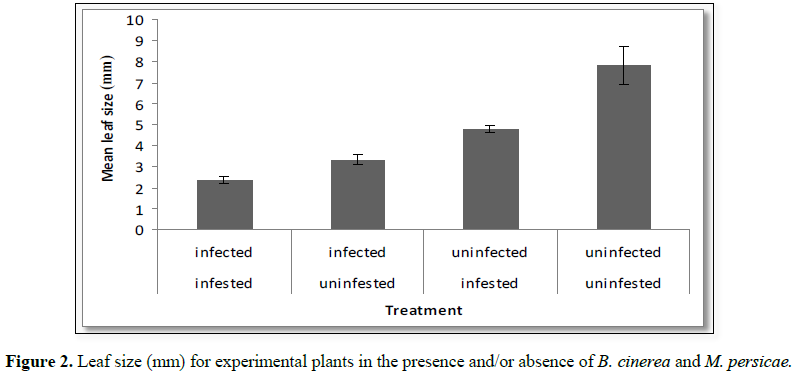
systemic B. cinerea and M. persicae on leaf size: There was a significant effect of aphid infestation
status and B. cinerea infection
status on the leaf size of the experimental plants (Table 1) where the presence of either resulted in a significant
reduction in leaf size. The interaction term was not significant (Figure 2).
Influence of
systemic B. cinerea and M. persicae on internode length: There was a significant effect of
aphid infestation status and level of B.
cinerea infection on the internode length of the experimental plants (Table 1) where the presence of either
resulted in a significant reduction in leaf size. The interaction term was not
significant (Figure 3).
Plant dry weight
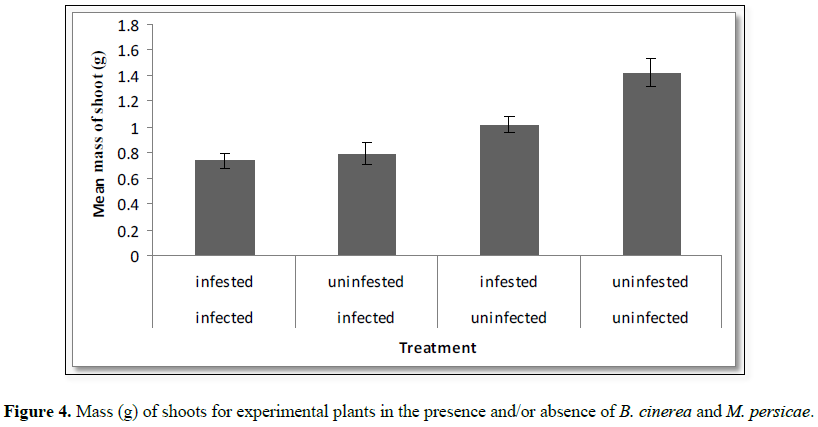
Influence
of systemic B. cinerea and M. persicae on dry shoot weight: A significant effect of aphid infestation status
and B. cinerea infection status on
the dry shoot weight of the experimental plants (Table 1) where the presence of either resulted in a significant
reduction in dry shoot weight. However, the interaction term was not
significant (Figure 4).
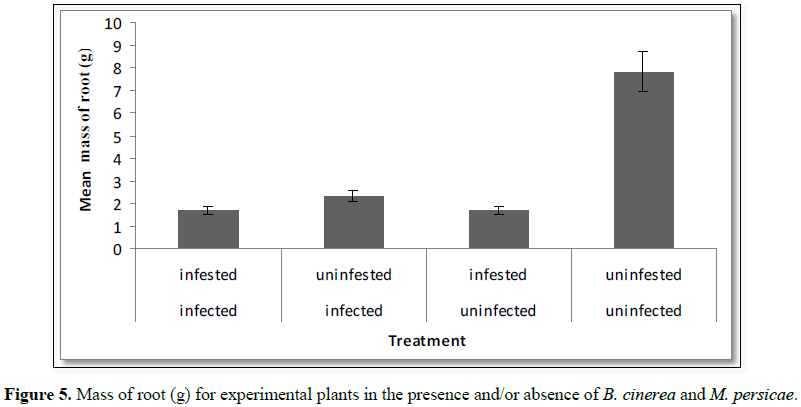
Influence of
systemic B. cinerea and M. persicae on dry root weight: Myzus persicae infestation or infection by B.
cinerea courses a significant decrease in the dry root weight of the plants
(Table 1). Because the primary
influence of either natural enemy was not additive, the interaction was
significant (Figure 5).
DISCUSSION AND
CONCLUSION
The result of the experiment showed that the
dynamics of the two natural enemies have an influencing factor on one another.
While lettuce plants infested with ten M.
persicae died within ten weeks of initial infestation, but B. cinerea infection did not result in
such mortality. Infection by B. cinerea
and infestation by M. persicae
reduced plant height, leaf size, internode length, plant dry weight. The rates
of expression of B. cinerea lesions
were however lower on aphid infested plants and aphid numbers were lower on
plants infected by B. cinerea. In
general it appears that the effect of aphid attack was greater than that of B. cinerea infection.
In a report by
Anna et al. [32] showed that following aphid’s infestation wastage of plants
and their produce occur as a result of injection of phytotoxin during feeding,
causing disintegration of chloroplast which decreases food production there by
slowing rate of plant growth. Despite the fact that the mechanism by which aphids affect plant
metabolism is not fully understood however, studies by Heil and Bostock [33],
Swarbrick et al. [34] and Golawska et al. [35] reported that the induction of defence is costly,
causing more need of assimilates by the plant. Also the ability of the
herbivore to manipulate the plant’s carbohydrate metabolism for its own use
deprives the plant sufficient carbohydrate for metabolic activities [36]. From
the result of the present study it is clear that plant height,
internode length, leaf size and dry shoot and root weights were significantly
reduced by the attack of M. persicae
and B. cinerea. Although the
interaction of aphid and B. cinerea
significantly reduced the root weight of the lettuce plants, the interaction of
aphid and B. cinerea did not
significantly affect plant height, internode length, leaf size, shoot weight
but their effect is exert independently
of one another.
Despite the resource quality of the lettuce
plant was not measured in this study. The result of the present study has found
that the effects of systemic B. cinerea
are detrimental to the increasing aphid population in lettuce plants. Infection
by systemic B. cinerea which spreads
into the plant tissues as the plant grows lowers the amount of nutrients
available, making the plant less favourable for the attacking aphid. In a
similar study on the effect of B. cinerea
and aphids (Rhodobium porosum
Sanderson) on rose plants (Rosa hybrida
L.). Mouttet et al. [37] reported that B.
cinerea induced the plant to synthesise secondary metabolites which either
have toxic effects, aversive and/or anti-feedant effects on aphids which may
subsequently attack. Such a negative relationship causes a reduction in the reproduction
rate of M. persicae, thereby lowering
its population size. This showed that co- interactions has the ability to
change the ecological interactions and spatial distribution of the insect
herbivore [37-41] in particular with regards to its interactions with its
natural enemies.
Result of the study showed that expression of
B. cinerea was drastically reduced
after infestation with M. persicae
when compared to non-infested plants. In a similar experiment Mouttet et al. [37]
found that infestation with aphids (Rhodobium
porosum Sanderson) on B. cinerea infected
plant result in lower expression of B.
cinerea and the infestation triggers the induction of Salicylic acid
(SA)-dependent pathway around the infection site which kill cells of B. cinerea and stop it growth. But,
continuous feeding by the aphids on the cell contents triggers the plant to
induce the wound-response pathways, (JA) and (ET) dependent pathways which
reduce the population of aphids [42-45].
In a related study
by Delucchi [46] on the effect of M.
persicae on the growth of its plant host, Brussels sprouts. A general
reduction in dry and fresh weight, height, internode length, leaf number and
size was reported, where the most striking observation was reduction in dry
root weight which was partly a due to the removal of assimilates, which were
otherwise available for storage. Brussels sprouts which were subjected to
probing and salivation by M. persicae
and on which continued feeding by the aphid was prevented showed exactly a
similar reduction accompanied by increased respiration of the plant.
Therefore,
Delucchi [46] as well as Mackauer and Way [47] concluded that the increased
respiration of the plant, a response to wounding and/or aphid salivation,
contributed considerably to a reduction in plant height, leaf size and dry
weight. However, finding by Heng-Moss et al. [48] reported that the reduction
in the plant fresh and dry weight occurs due to the reduction of photosynthesis
in leaves which have been injured by interaction of aphid infestation and
pathogen attack due to an increased synthesis of defensive chemicals in
response to the attack [49].
The present research has confirmed that
co-interactional relationship between a systemic pathogen and an insect herbivore attacking lettuce plants reduces
growth of the lettuce plant [50,51]. Therefore, the results of the experiment
shows that B. cinerea affect aphid
and also aphid affect B. cinerea and
each of them stress the lettuce plant. In addition, the experiment confirmed
the existence of a negative relationship between M. persicae and B. cinerea
where they independently stress the host plant, and in addition to the
reduction of the population growth rate of each other, they also reduce the
growth rate of the host plant, probably by triggering the induction of defence
chemicals by the plant at the expense of other important vital functions
[52-55]. This shows that B. cinerea
may have far-reaching effects on coexisting insect
herbivores. Therefore this research will serve as a
valuable pointer in increasing our understanding of the ecological consequences
of a ubiquitous but hitherto understudied interaction [56,57].
1. Mackean DG (2004) Aphids,
Hemiptera, life cycle, behavior reproduction and feeding damage to plants and
control of damage. Available at: http://www.biology-resources.com/aphid-01.html
2. Agrios NG (2005) Plant Pathology.
Elsevier Academic Press, USA.
3. Yahaya SM, Ahmed I (2015) Does
seed serve as inoculum source for systemic Botrytis
cinerea infection in lettuce. India J Plant Sci 4: 7-21.
4. Blackman RL, Eastop VF (2000)
Aphids on the world’s herbaceous plants and shrubs. Wiley, Chichester, England.
5. Yahaya SM, Mardiyya AY (2019)
Review of post-harvest losses of fruits and vegetables. Biomed J Sci Tech Res
13.
6. Michael JS, Jennifer ST, Bart PHJT
(2005) Plant mediated interactions between pathogenic microorganisms and
herbivorous arthropods. Annu Rev Entomol 51: 663-689.
7. Hua L, Yong C, Zhaquan Z, Boqiang
L, Guozheng Q, et al. (2018) Pathogenic mechanism and control strategies of Botrytis cinerea causing post-harvest
decay in fruits and vegetables. Food Qual Saf 3: 111-119.
8. Samir D, Amnon L (2007)
Post-harvest Botrytis infection: Etiology, development and management. In Elad
Y, Botrytis: Biology, pathology and control. Springer, Dordrecht, the
Netherlands, pp: 349-362.
9. Govrin EM, Levine A (2000) The
hypersensitive response facilitates plant infection by the necrotrophic pathogen
Botrytis cinerea. Curr Biol 10:
751-757.
10. Elad Y, Stewart A (2004) Microbial
control of Botrytis cinerea. In Elad Y, Botrytis: Biology, Pathology and
Control. Springer, Dordrecht, the Netherlands, pp: 223-236.
11. Yahaya SM, Fagwalawa LD, Lawan M
(2015) Influence of airborne and seed inoculum in the initiation of leaf, stem
and root infection by systemic Botrytis
cinerea in Lettuce (Lactuca sativa). J Plant Pathol
Microbiol 11.
12. Williamson B, Tudzynski B,
Tudzynski P, Van Kan JAL (2007) Botrytis
cinerea: The cause of grey mould diseases. Mol Plant Pathol 8: 561-580.
13. Holz G, Coertze S, Williamson B
(2004) The ecology of Botrytis on plant surfaces. In Elad Y, Botrytis: Biology,
pathology and control. Springer, Dordrecht, the Netherlands, pp: 9-27.
14. Van Kan JAL (2005) Infection
strategies of Botrytis cinerea. Acta
Horticulturae 669: 77-89.
15. Sowley ENK (2006) Epidemiology of Botrytis cinerea. PhD Thesis, University
of Reading, UK.
16. Elias SNK, Shaw MW, Dewey FM
(2010) Persistent symptomless, systemic and seed-borne infection of lettuce by Botrytis cinerea. Eur J Plant Pathol
126: 61-71.
17. Yahaya SM, Mardiyya AY, Sakina SB,
Hayatu LW (2019) Disease cycle and infection strategies of systemic plant
pathogen Botrytis cinerea. Novel Res
Microbiol J 3: 204-214.
18. Norman JC (1992) Tropical
vegetable crops. Stockwell Ltd., New York, pp: 13-53.
19. Ramu S, Kuppusamy P, Lingan R,
Gandi K (2011) Efficacy of different biological control agents against major
post-harvest pathogens of grapes under room temperature storage conditions. Phytopathology
50: 55-65.
20. Yahaya SM, Fagwalawa LD, Ali MU,
Umma M, Lawan M, et al. (2016) Symptomless transmission of seed-borne Botrytis cinerea into the seed of next
generation lettuce plant (Lactuca sativa).
J Plant Sci Res 1.
21. Palumbo JC (2001) Influence of
admire and platinum on the population growth of the lettuce aphid under field
condition. In Byrne DN and Baciewicz P. (ed.) Vegetable Report Series.
University of Arizona, College of Agriculture and Life Science, pp: 14-19.
22. Nicolaos EM, Dimitris EG (2002)
Fungal and bacterial disease. In Emerging Infectious Diseases. Kluwer Academic
Publishers: New York, pp: 34-46.
23. Barbosa P (1991) Plant pathogens
and non-vector herbivores. In: Barbosa P, Krischik VA and Jones CG (ed.).
Microbial Mediation of Plant-Herbivore Interactions. Wiley, New York, pp:
341-382.
24. Hatcher PE, Paul ND, Ayres PG,
Whittaker JB (1994b) The effect of foliar disease (rust) on the development of Gastrophysa viridula (Coleoptera:
Chrysomelidae). Ecol Entomol 19: 349-360.
25. Stout MJ, Thaler JS, Thomma BPJ
(2006) Plant-mediated interactions between pathogenic microorganisms and
herbivorous arthropods. Annu Rev Entomol 51: 668-689.
26. Rodger G, Rahier M, Naisbit RE
(2007) Coping with an antagonist: the impact of a phytophatogenic fungus on the
development and behavior of two alpine leaf beetles. Oikos 116: 1514-1523.
27. Kruess A (2002) Indirect
interaction between a fungal plant pathogen and a herbivorous beetle of the
weed Cirsium arvense. Oecologia 130:
563-569.
28. Cory JS, Hoover K (2006)
Plant-mediated effects in insect-pathogen interactions. Trends Ecol Evol 21:
278-286.
29. Dixon AFG (1985) Structure of
aphid populations. Annu Rev Entomol 30: 155-174.
30. Hilton A, Armstrong R (2006)
Post-hoc Tukey ANOVA test. Available at: http://eprints.aston.ac.uk/93317/1/
31. Rehman MK (2013) Problem solving
and data analysis using Minitab: A clear and easy guide to six sigma
methodology. Wiley, Available at: http://www.ebookee.org/Problem-Solving-and-Data-Analysis-9
32. Anna MB, Leon V, Carlos SJ,
Francois N, Burger V (2012) Phloem feeding insect stress and photosynthetic
gene expression. In: Najafpour M. (ed.) Applied photosynthesis. Springer: USA,
pp: 22-33.
33. Heil M, Bostock RM (2002) Induced
systemic resistance (ISR) against pathogens in the context of induced plant defenses.
Annu Bot 89: 503-512.
34. Swarbrick PJ, Schuze-Lefert P,
Scholes JD (2006) Metabolic consequences of susceptibility and resistance
(race-specific and broad-spectrum) in barley leaves challenged with powdery
mildew. Plant Cell Environ 29: 1061-1076.
35. Golawska S, Kryzyzanowski R,
Lukasik (2010) Relationship between aphid infestation and chlorophyll content
in Fabaceae species. ACTA Biologica Botanica 52: 76-80.
36. Rodriguez-Soana C, Law BR, Rufus I
(2011). Manipulation of natural enemies in agroecosystems habitat and
semiochemicals for sustainable insect pest control. In Larramendy ML and
Soloneski S (ed.). Integrated Pest Management and Control Current and Future
Tactics. InTech, pp: 89-126.
37. Mouttet R, Philippe B, Thomas C,
Desneux N (2011) Phytophagous arthropods and a pathogen sharing a host plant.
PLoS One 6: e18840.
38. Paul ND, Hatcher PE, Taylor PE
(2000) Coping with multiple enemies: An integration of molecular and ecological
perspectives. Trends Plant Sci 5: 220-225.
39. Ohgushi T (2005) Indirect
interaction webs: herbivore-induced effects through trait change in plants. In
Ohgushi T, Schmitz O and Holt RD (ed.) Ecological Reviews. Trait-Mediated
Indirect Interactions. Cambridge University Press, p: 183.
40. Viswanathan DV, McNickle G, Thaler
JS (2008) Heterogeneity of plant phenotypes caused by herbivore-specific
induced responses influences the spatial distribution of herbivores. Ecol
Entomol 33: 86-94.
41. Mauck KE, De Morales CM, Mescher
MC (2010) Effects of cucumber mosaic virus infection on vector and non-vector
herbivores of squash. Communicative and Integrative Biology 3: 579-582.
42. Kanno H, Fujita Y (2003) Induced
systemic resistance to rice blast fungus in rice plant infested by white-backed
plant hopper. Entomologia Experimentalis et Applicata 107: 155-158.
43. De vos M, Van Zaanen W, Koornef A,
Korzelius JP, Dicke M (2006) Herbivore-induced resistance against microbial
pathogens in Arabidopsis. Plant Physiol 142: 352-363.
44. Hatano E, Kunert G, Michaud JP,
Wolfgang W, Weisser W (2008) Chemical cues mediating aphid location by natural
enemies. Eur J Entomol 105: 797-806.
45. Wassim EC, Roy K, Tatyana S,
Heather R, Florence N, et al. (2008) Distinct roles of jasmonates and aldehydes
in plant-defense response. PLoS One 3: e1904.
46. Delucchi VL (1976) Studies in
biological control. International Programme, Cambridge University Press 9:
414-420.
47. Mackauer M, Way MJ (1976) Myzus
persicae Sulzer an aphid of world importance. In Delucchi VL (ed.). Studies in
Biological Control. University Press: Cambridge, UK, pp: 51-119.
48. Heng-Moss TM, Macedo T, Markwell
JP, Baxebdale FP, Quisenberyy SS, et al. (2003) Comparison of chlorophyll and
carotenoid concentrations among Russian wheat aphid (Homoptera: Aphididae) -
infested wheat isolines. J Econ Entomol 96: 475-481.
49. Blackman RL (1974) Life cycle
variation of Myzus persicae (Sulzer)
(Homoptera: Aphidiidae) in different parts of the world, in relation to
genotype and environment. Bull Entomol Res 63: 595-609.
50. Dixon AFG (1998) Aphid ecology: An
optimization approach. Chapman and Hall: London, pp: 2-26.
51. Eastop VF (1986) Aphid plant
associations. Co-evolution and Systematics. Clarenden Press: Oxford, pp: 33-54.
52. James WC (1974) Assessment of
plant diseases and losses. Ann Rev Phytopathol 12: 27-48.
53. Margaritopoulos JT, Tzortzi M,
Zarpas KD, Tsitsipis JA, Blackman RL (2006) Morphological discrimination of Aphis gossypii (Hemiptera: Aphididae)
populations feeding on Compositae. Bull Entomol Res 96: 153-165.
54. Minitab Inc. (2009) Minitab
statistical software, Release 16 for Windows, State College, Pennsylvania.
55. O’Neil TM, Gladders P, Ann DM
(1997a) Prospects for integrated control of lettuce diseases. In: Crop
Protection and Food Quality Meeting Customer Needs. Proceedings of BCPC/ANPP
Conference. University of Kent, UK, p: 485.
56. Palumbo JC, Mullis J, Reyes F,
Amaya A (1998) New insecticide alternatives for aphid management in head
lettuce. In Oebker NFE (ed) Vegetable Report Series. University of Arizona.
College of Agriculture and Life Science, pp: 1-10.
57. Synder W, Ives A (2003)
Interaction between specialist and generalist natural enemies: Parasitoids,
predators, pea aphids biocontrol. Ecology 84: 91-97.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Genetics and Cell Biology (ISSN:2639-3360)
- Advances in Nanomedicine and Nanotechnology Research (ISSN: 2688-5476)
- Journal of Microbiology and Microbial Infections (ISSN: 2689-7660)
- Journal of Genomic Medicine and Pharmacogenomics (ISSN:2474-4670)
- Journal of Veterinary and Marine Sciences (ISSN: 2689-7830)
- Proteomics and Bioinformatics (ISSN:2641-7561)
- Food and Nutrition-Current Research (ISSN:2638-1095)