2420
Views & Citations1420
Likes & Shares
The determination of plasma L-dopa was performed by a capillary electrophoresis system (HP-3DCE Agilent, Waldron, Germany); the samples were filtered in an acetate cellulose membrane (0, 22mm) and injected directly on silica capillary. The injections were in the hydrodynamic mode in silica capillary (52cm x 75mm ID; Agilent), and the pressure was 50mBar. The run voltage applied was 10kV. The runs were performed in borate buffer 50mM, pH 9.0. L-dopa was detected at 236 nm using a Diode Array Detector. Pure L-dopa was used as standard to compare migration time and UV spectra.
RESULTS AND DISCUSSION
CE is well suited for diagnostic analysis, been used to determination of forensic analysis, due its applicability includes analysis of drugs, serum proteins, hemoglobin variants, and others molecules [6].
The importance of measuring serum L-dopa consists in the possibility to monitor the PD-patient that can present a poor absorption, depending of his food habits, and the time of the disease progression.
The maximum absorption of L-dopa is about 200nm, although this wavelength has more possibility of interferences from serum peptides, small protein, or other unknown compounds. The Figure 1 shows the L-dopa spectra, with no spectral features in the visible region, but with two others peaks at 236 and 285nm that can be used during analysis to decrease the interfering possibility.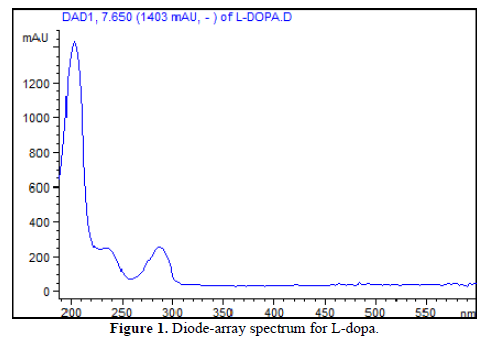
Using a run buffer of 50mM borate of pH 9.0 facilitated a rapid analysis time, due its negative charge net of unproton carboxylic group, with migration time of 5.4 min (Figure 2). The L-dopa structure is shown in Figure 3.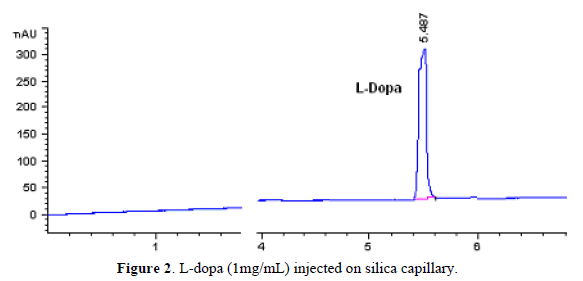
A linear detector response was obtained when decreasing concentrations of L-dopa were injected in the silica capillary.
Figure 4 show a rat plasma CE profile after protein precipitation, carbidopa and L-dopa were detected separately. The L-dopa spectrum obtained was consistent with the previously found after standard injection (Figure 1). The L-dopa plasma concentration average was 5.44±0.37mg/mL.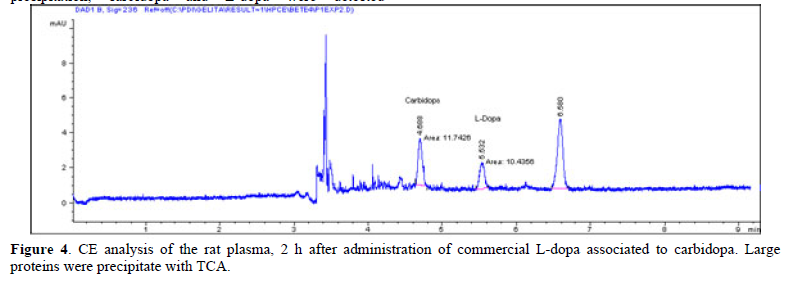
Tesoro [6] made an overview of methods to extract and analyze L-Dopa, including an electrophoresis capillary method; however the measurements are done in conditions that extract this analyte since plant matrice.
The method preliminary developed in this study is simple, rapid, and suitable for the quantification of L-dopa or carbidopa in plasma or other biological fluid samples from clinical, pharmacological or therapeutic studies. In additional, diode array detection can reduce doubts about analytes nature, because in L-dopa oxidation several intermediate products are formed.
ACKNOWLEDMENTS
The authors tank Dr. Jaime Amaya-Farfán, Faculty of Food Engineering of the State University of Campinas-SP, Brazil, for provide the capillary electrophoresis apparatus.
- Cenci MA, Crossman AR (2018) Animal models of L-dopa-induced dyskinesia in Parkinson's disease. Mov Disord 33(6): 889-899.
- Fabbrini A, Guerra A (2021) Pathophysiological mechanisms and experimental pharmacotherapy for L-dopa-induced dyskinesia. J Exp Pharmacol 29(13): 469-485.
- Jiang R, Yang J, Mei S, Zhao Z (2022) Determination of levodopa by chromatography-based methods in biological samples: A review. Anal Sci 38: 1009-1017.
- Pang SYY, Ho PWL, Liu HF, Leung CT, Li L, et al. (2019) The interplay of aging, genetics and environmental factors in the pathogenesis of Parkinson’s disease. Transl Neurodegener 8: 23.
- Puris E, Gynther M, Auriola S, Huttunen KM (2020) L-Type amino acid transporter 1 as a target for drug delivery. Pharm Res 37: 88.
- Tesoro C, Lelario F, Ciriello R, Bianco G, Capua DA, et al. (2022) An overview of methods for L-dopa extraction and analytical determination in plant matrices. Separations 9:





