Research Article
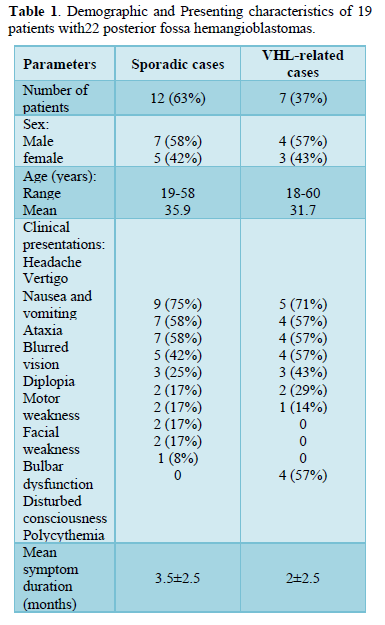
Posterior Fossa Hemangioblastoma: Clinical Features and Surgical Outcome in Retrospective Series of 19 Cases
4391
Views & Citations3391
Likes & Shares
Background Data: Hemangioblastoma represents about 7%-12% of posterior fossa tumors in adults which occur either sporadically (60%-75%) or in relation to von Hippel-Lindau disease (VHL) (25%-40%). Although, stereotactic radiosurgery (Gamma Knife radiotherapy) has been suggested as an effective treatment, microsurgical resection remains the definitive treatment.
Purpose: The purpose is to analyze the clinical and imaging characteristics of posterior fossa hemangioblastomas, rates of surgical excision and clinical outcome.
Study Design: Retrospective observational analytic study.
Patients and Methods: This is an analytical retrospective study enrolled 19 patients (11 males and 8 females; with mean age of 35.9 years in sporadic cases and 31.7 years in VHL related cases) with posterior fossa hemangioblastomas who underwent microsurgical resection of 22 lesions through a midline or lateral suboccipital approach.
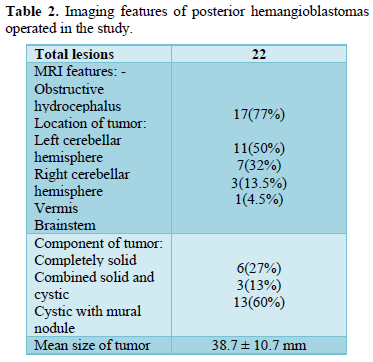
Results:12 cases (63%) have sporadic posterior fossa hemangioblastoma and the other 7 cases (37%) had clinical confirmation of VHL disease from the same Bedouin Sinai family. The most common presenting symptoms and signs were related to increased intracranial pressure (ICP) followed by cerebellar signs. There were 6 (27%) completely solid, 3 (14%) combined solid and cystic, and 13 (59%) cystic tumors with mural nodule. Gross total resections were achieved in 18 operations (82%), partial resections in 4 lesions (18%). Favorable outcomes were achieved in 14 patients (74%), fair outcome in 3 patients (16%) and death in 2 patients (10%).
Conclusion: Posterior fossa hemangioblastomas are usually cerebellar lesions and reach considerable size by the time of diagnosis. The symptoms depend on the tumor-associated cysts, and lesion growth rate. Microsurgical resection is the treatment of choice for sporadic HBLs, those associated with VHL disease, and those showing growth on repeated imaging. The surgical technique involves circumferential lesion dissection, always attempting en-bloc resection to avoid intraoperative bleeding. Our favorable outcomes suggest that microsurgical resection is the treatment of choice for hemangioblastomas of the posterior fossa. Patients with VHL disease should be followed closely for a long-term period, as delayed lesion growth may occur.
Keywords: Posterior fossa, Hemangioblastoma, Outcome, Recurrence, Von Hippel-Lindau disease
INTRODUCTION
Hemangioblastoma is a benign, slowly growing, highly vascular, (WHO Grade I) neoplasms that occur in any part of the CNS. This tumor accounts for 1.5%-2.5% of all intracranial tumors and 7%-12% of posterior fossa tumors in adults [1]. They mainly arise in the cerebellar hemispheres (76%), making them the most common primary neoplasm of the cerebellum in adults [2]. Hemangioblastomas may appear solid, solid-cystic, or mainly cystic with a small mural nodule (nidus) and the nodule of hemangioblastomas is superficially located and typically abuts a pial surface and cells and abundant capillaries [3]. They occur either sporadically (60%-75% of cases) or in the context of von Hippel-Lindau disease (VHL) (25%-40%), an autosomal dominant neoplastic syndrome affecting multiple organ systems [1]. They are believed to appear more often in males than in females with symptoms typically occur earlier in VHL patients (30-40 years) compared with sporadic patients (40-50 years) [4]. Clinical findings associated with CNS hemangioblastomas are due to anatomically specific mass effect related to the tumor itself, peritumoral cyst, and/or a peritumoral edema with no specific signs or symptoms for hemangioblastoma. Patients with peritumoral cysts typically develop signs and symptoms as a result of the rate of cyst growth and its volume so microsurgical gross total resection is considered a curable measure [5]. Herein, we collected clinical data retrospectively of 19 cases of posterior fossa hemangioblastomas and described the management experience.
AIM OF THE WORK
Our aim was to study and analyze the clinical characteristics of posterior fossa hemangioblastomas, imaging features, rates of surgical resection, postoperative complications and their rates, mortality, and clinical outcomes. In addition, to find out whether there is a difference in these parameters between sporadic and VHL related posterior fossa hemangioblastomas.
PATIENTS AND METHODS
Study design: The current study is a retrospective observational analytical study of a total 19 patients with posterior fossa hemangioblastomas who underwent surgical excision in the period between 2012-2015. Cases enrolled in this study were selected based on eligibility criteria which included inclusion criteria; histologically and radiologically confirmed cases of posterior fossa hemangioblastomas (WHO Grade I) whether sporadic or VHL-related which underwent microsurgical resection and excluding cases who managed expectantly or by Gamma Knife radiotherapy.
Data collection: patients’ data were collected and retrospectively reviewed after approval by the Institutional Review Board of our hospital. These data comprise patients’ demographics, presenting clinical symptoms and signs, imaging features, surgical details and postoperative outcomes. The age and gender of patients were analyzed and the medians, ranges, means, and standard deviations were calculated, as well as the presenting symptoms and clinical signs in which descriptive statistics were used to analyze. Tumor location within posterior fossa was identified either in the cerebellum; brainstem; cerebellopontine angle; the fourth ventricle or craniometrical junction. Tumor morphology was analyzed as solid, cystic or mixed. The surgical parameters analyzed were the following: the extent of tumor resection; intraoperative blood loss; postoperative complications; and length of hospital stay. The extent of resection was listed as total, subtotal/near total or partial. Outcomes were categorized as favorable (no postoperative neurologic deficits and no postoperative complications); fair (mild postoperative neurologic deficit or postoperative complications); poor (debilitating postoperative complications or grave neurologic deficits); and death.
Imaging Evaluation: Patients were evaluated preoperatively with brain high-resolution magnetic resonance imaging (MRI), and some patients also underwent computed tomography angiography, magnetic resonance angiography, or digital subtraction angiography. MRI with T1/T2-weighted imaging and gadolinium enhanced sequences were used to demonstrate the location, size, and texture of the tumors, and the angiography was performed to evaluate the feeding arteries and draining veins. The tumor size was defined as the largest diameter in all three coordinate planes of the lesion, including the extra tumoral cyst.
Surgical Technique: A total of 22 surgical procedures in 19patients were done for microsurgical tumor debulking (3 patients with VHL had multiple cerebellar hemangioblastoma, each patient of them operated twice). The appropriate surgical approach to the tumor was selected with consideration to the position of the mass, presence or absence of a large cystic component, secondary hydrocephalus and surrounding edema. The midline or ipsilateral suboccipital approaches were done according to the site of the lesions either located in the cerebellar hemisphere, vermis and dorsal medulla. Tumors were identified intraoperatively by microscopy with assistance of intraoperative ultrasonography in some cases especially those with large cystic component to localize hyperechoic mural nodule and hypoechoic cysts. They were superficial racemose hemangiomas appearing bright (orange) or dark red. We proceeded under the principle that the feeding arteries should be divided in advance and the draining veins should be ligated last by using low voltage bipolar coagulation. The blood supply to the tumor was interrupted before the resection of the tumor, and then complete tumor resection was carried out after peripheral tissue dissection around the tumor margin avoiding resection inside the tumor. Finally, the draining vein was ligated. As internal decompression of tumors may cause uncontrollable bleeding, en bloc resection was carried out even if the tumor was relatively large. For cystic hemangioblastomas, the mural nodule is excised micro surgically and the peritumoral cyst walls are left undisturbed. None of the cases enrolled in this study underwent preoperative embolization.
Postoperative follow up: Data concerning early postoperative hospital period, hospital stay, postoperative complications and follow up MRI imaging were collected and analyzed.
RESULTS
Patients’ demographics: There were 11 males (58%) and 8 females (42 %) with age at time of surgery ranging from 18-60 years with mean age of 35.9 years in sporadic cases and 31.7years in VHL related cases. These cases were 12 cases (63%) of sporadic posterior fossa hemangioblastoma in whom 7 were males (58% of sporadic, 36% of total) and 5 were females (42% of sporadic, 26% of total). The other 7 cases (37%) had clinical confirmation of VHL disease. They were series of 7 cases (4 males and 3 females), from the same Bedouin Sinai family, who were managed in departments of neurosurgery, urology and ophthalmology during the previous 2 decades with age at surgery (18,25,26,28,30,35,60) years. 3 patients harbored retinal hemangioblastomas, 1 patient harbored pancreatic carcinoma, 1 patient had spinal hemangioblastoma, and 3 had multiple cerebellar hemangioblastoma.
Presenting symptoms and clinical signs: The most common presenting symptoms and clinical signs were related to increased intracranial pressure (ICP) followed by cerebellar signs. The most frequent preoperative symptoms in sporadic cases were headache reported in 9 patients (75%), vertigo in 7 patients (58%), nausea and vomiting in 7 patients (58%), ataxia in 5 patients (42%), blurred vision in 3patients (25%), diplopia in 2 patients (17%), motor weakness in 2 patients (17%), facial weakness in 2 patients (17%), Bulbar dysfunction in 2 patients (17%) and disturbed conscious level in one patient (8%). The most frequent preoperative symptoms in VHL related cases were headache reported in 5 patients (71%), vertigo in 4 patients (57%), nausea and vomiting in 4 patients (57%), ataxia in 4 patients (57%), blurred vision in 3 patients (43%) and diplopia in 2 patients (29%).
The duration of symptoms before diagnosis ranged from two months to thirteen months with mean duration of symptoms was 3.5±2.5 months in sporadic cases and 2±2.5 months in VHL related cases.
Patients with VHL were presented in addition with associated pathologies; paraparesis with sensory level in one patient (14%) who had spinal hemangioblastoma. Polycythemia was documented in 4 cases (57%) of them. Patients’ characteristics and their presenting symptoms are detailed in Table 1.
Tumor characteristics and imaging features analysis (Table 2)
There were 6 (27%) completely solid, 3 (13%) combined solid and cystic, and 13 (60%) primarily cystic tumors with mural nodule. The MRI scans showed a tumor in the left cerebellar hemisphere in 11 (50%) patients, right cerebellar hemisphere in 7 (32%), vermis in 3 (13.5%) and brainstem (dorsal medulla) in 1 patient (4.5%). The total size of the tumor range from 20-52mm was mean of 38.7 ± 10.7 mm. The presence of obstructive hydrocephalus was identified in

17 (77%) patients. As regard of VHL related cases, there were 10 lesions in 7 cases, 6 were cystic with mural nodule and 4 cases were solid with MRI showing 8 lesions in the cerebellar hemispheres, 1 lesion in vermis and 1in brainstem.
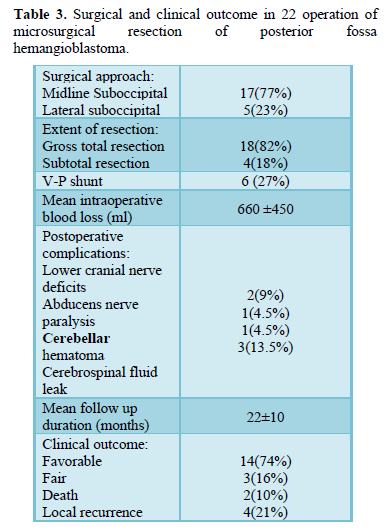
Surgical characteristics (Table 3)
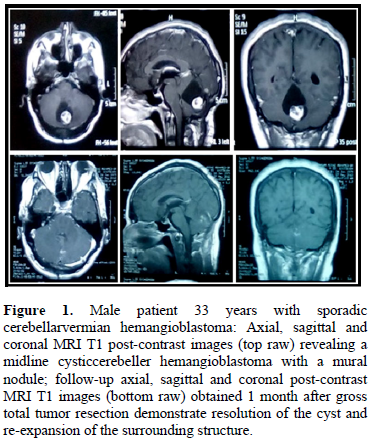
Preoperative embolization was not performed in any of the cases enrolled in our study. Lateral Suboccipital approach was applied in 17 procedures (77%) and midline suboccipital approach was applied in 5 procedures (23%). Gross total microsurgical resections were achieved in 18 operations (82%) (Figure 1). Partial microsurgical resections were performed in 4 lesions (18%) because massive tumor bleeding was encountered intraoperatively and tight adhesion between the tumor and surrounding tissues. Six patients underwent adjunctive ventriculoperitoneal shunting due to associated secondary

hydrocephalus. The mean blood loss reported was 660 ±450 ml.


Clinical outcomes and complications (Table 3)
Out of 22 microsurgical procedures performed for 19 patients, favorable outcomes were achieved in 14 patients (74%), fair outcome in 3 patients (16%) and death in 2 patients (10%). Three patients with fair outcome were still suffered from mild degree of motor weakness that not improved on physiotherapy and patients were still unsatisfied by the results. Four patients experienced improvement presented with a new-onset symptom resulting from recurrent hemangioblastomas during the follow up at mean of 14±10 months after the primary surgery, which involved 1 case in sporadic and the other 3 in VHL-related hemangioblastomas, respectively. Symptoms improved without permanent neurological deficits after the second operation. One patient underwent a subsequent operation for removal of spinal hemangioblastoma 2 years after the first surgery. Two patients with VHL disease who presented with multiple intracranial hemangioblastomas experienced tumor progression during the study.
Postoperative complications included lower cranial nerves deficits in 2 patients, abducent nerve paralysis in 1 patient, postoperative cerebellar hemorrhage in 1 and cerebrospinal fluid (CSF) leak in 3 patients. Lower cranial nerve deficits including swallowing difficulty and hoarseness were all recovered spontaneously in the follow-up. One case showed abducent nerve paralysis which failed to recover completely. One patient suffered from postoperative cerebellar hemorrhage and underwent an emergency operation to evacuate the hematoma, and the patient recovered favorably after surgery. CSF leak in 3 patients was successfully treated with lumbar drainage for 5 days. Death occurred in 2 patients due to non-neurosurgical issues during postoperative ICU period, one patient due to severely hospital acquired pneumonia and the second patient due to unfortunately pulmonary embolism secondary to lower limb deep venous thrombosis (DVT).
DISCUSSION
In this case series, we analyzed the results of microsurgical resection of posterior fossa hemangioblastoma including patients with the sporadic type and those with VHL related disease to gain insight into the features and reasonable management of these tumors. The average age of the patients at first presentation of cerebellar hemangioblastomas was ranging from18-60 years with mean age of 35.9 years in sporadic cases and 31.7 years in VHL related cases which was compatible with the previous findings reported by researcher’s [3,7,8]. We noted that the 7 patients of VHL related hemangioblastomas who were included in this study (37%) presented at an earlier age than do sporadic cases who tended to present at an older age and these data was correlated with results reported by researcher [1].
All patients were symptomatic at diagnosis, and symptoms due to increased intracranial pressure or a cerebellar mass effect were most common. This could be explained by the benign nature of hemangioblastoma with slow rate of growth tending to compress rather than infiltrate the surrounding tissues and thus remain silent until reaching a volume sufficient to produce prominent regional pressure in the posterior fossa. In this study, the mean diameter of hemangioblastomas was 38.7 ± 10.7 mm. And hence, it was not surprising that hydrocephalus was identified in the majority of 17 patients (77%) because of the mass effect of the tumor with obstruction of the fourth ventricle. This was consistent with the results reported by researcher’s [1,8]. It was noted that patients with sporadic lesions presented much late than those with VHL-related tumors. This may be due to VHL patients or their family’s members are evaluated for the presence of hemangioblastomas of the entire neuroaxis, unlike sporadic cases are usually diagnosed by the time of symptoms [1]. Researcher suggested that differences in the molecular and genetic origins of hemangioblastomas could be another possible explanation [1].
Among the 22-posterior fossa hemangioblastomas in this study, up to 18 (82%) were located in cerebellar hemisphere. This was similar to other studies which all reported that the cerebellar hemisphere to be the most common location of posterior fossa hemangioblastoma [1-3,7,9]. Up to 13 lesions (60%) were well circumscribed cystic lesions with hyper vascular mural nodules similar to the results reported by researcher’s [10,11]. Although there are no histological differences between the cystic and solid tumor subtypes, solid subtypes may require a more extended approach to achieve an adequate operative space [12, 13]. Researchers characterized the biochemical composition of intra-tumoral and peritumoral cysts in hemangioblastomas, and found that the content was highly similar to serum and not to tumor. Their results favor the theory that hemangioblastoma-associated cysts are caused by abnormal vascular leakage [14].
Although this study did not demonstrate the natural history or growth patterns of posterior fossa hemangioblastomas, in most lesions, the cyst was larger than the solid tumor at diagnosis supporting what be reported by researchers. that the space of enlargement was much faster for cysts than for solid hemangioblastomas. Thus, this result reasonably interpreted that an associated cyst of the mass effect-producing tumors was commonly and primarily responsible for the symptoms or even life-threatening morbidity due to the cyst expansion [15]. As a result, the importance of cysts in the evolution of these tumors to a symptom-producing stage has to be considered during therapeutic planning. Because stereotactic radiosurgery does not quickly reduce the mass effect, it may not be effective for treating the large symptomatic cystic cerebellar hemangioblastomas in contrast to surgical resection of cerebellar hemangioblastomas which is a timely treatment, and is the more favored treatment of choice in those patients [6].
In our study, complete microsurgical resection of the tumors could be achieved in 18 lesions (82%) of posterior fossa hemangioblastomas with favorable outcomes that achieved in 14 patients (74%) and the incidence of perioperative morbidity or mortality was low. In addition, the majority of patients benefited immediately from surgical removal of cerebellar hemangioblastoma. Even in four lesions (18%) that underwent partial tumor resection because of massive bleeding, there were no new cerebellar-related signs and symptoms or exacerbation of preexisting symptoms. Our experience confirms the validity of the concept that surgery is an effective and safe strategy, and prevents progressive functional decline in patients with posterior fossa hemangioblastomas. Researcher’s also report that surgery of intracranial sporadic hemangioblastoma’s provides good quality of life, and multimodal therapeutic approach, including radiosurgery, is reserved for the specific situations, such as inaccessible brainstem hemangioblastoma or recurrent hemangioblastoma [16].
With advances in the knowledge of hemangioblastomas and surgical equipments such as operating microscope and establishment of elegant surgical technique, most cerebellar hemangioblastomas can be managed by gross total resection, with less complications and improved outcomes as measured by functional recovery. In our experience, en-bloc resection is best to prevent hemorrhage, which may arise as a result of debulking maneuvers. We proceeded in an AVM-like manner under the principle that the feeding arteries should be divided in advance and the draining veins should be ligated last by using low voltage bipolar coagulation and then complete tumor resection was carried out after peripheral tissue dissection around the tumor margin. En bloc resection was carried out even if the tumor was relatively large. The tumor-feeding arteries are often located deeply on both sides of the tumor, while the large draining veins usually arise from the tumor surface. With careful observation, these veins can be identified accurately during the operation. For cystic hemangioblastomas, the mural nodule is excised micro surgically and the cyst walls are left undisturbed considering the added morbidity of dissecting a cyst away from adjacent brain and excised only in cases in which the presence of tumor tissue is confirmed by cyst wall enhancement in preoperative MRI brain. This technique was also adopted by researcher’s [3,7,10,17,18].
In addition, none of the cases enrolled in this study had undergone preoperative embolization which is actually considered to be one of our study limitations. On the other hand, this can be regarded as a real challenging for en bloc resection technique to achieve good surgical outcome alone. Moreover, there are potential risks and complications associated with preoperative embolization that cannot be ignored. Researcher’s [19] reported that in 3 patients with cerebellar hemangioblastomas who underwent preoperative embolization, the outcome was unfavorable and acute tumor bleeding and death occurred in all of them. Apart from the acute bleeding complications, tumor swelling and vessel occlusion with consecutive infarction are described as well [20,21].
Previous studies have reported recurrence rates of 20–33% in patients with sporadic hemangioblastomas and in those with VHL diseases after resection of hemangioblastomas of the central nervous system [3,5,7,22]. Our data showed four patients (21%) were presented by recurrent posterior fossa hemangioblastomas during the follow up at mean of 14±10 months after the primary surgery, which involved 1 case in sporadic and the other 3 in VHL-related hemangioblastomas, respectively.
For sporadic central nervous system hemangioblastomas, researchers. suggested an MRI scan of the involved neuronal axis at 6 and at 12–24 months in the postoperative period. Other studies conclude that long-term imaging follow-up is needed to detect late recurrences of hemangioblastomas, even if the clinical course is benign after total removal of the tumor [10,11].
In our series, partial resections were performed in 4 lesions (18%) because massive tumor bleeding was encountered intraoperatively and tight adhesion between the tumor and surrounding tissues, these cases were managed by adjuvant Gamma Knife radiotherapy with good control of tumor progression as showed on follow up MRI. Researcher’s [6] analyzed 26 intracranial hemangioblastomas of which 22 tumors were residual from previous surgical resection; they showed that gamma knife radiosurgery offers a reasonable rate of tumor control. On the other hand, other studies suggested that in the presence of symptomatic residual tumor at follow-up, it is preferable to carry out secondary resection which reporting favorable outcome in their experience with secondary surgery in spite that the tissue plane between the tumor margin and cerebellum may be ill-defined due to postoperative adhesion [2,3,9].
CONCLUSION
Posterior fossa hemangioblastomas are usually cerebellar lesions and reach considerable size by the time of diagnosis. The symptoms depend on the tumor-associated cysts, and lesion growth rate. Microsurgical resection is the treatment of choice for sporadic HBLs, those associated with VHL disease, and those showing growth on repeated imaging. The surgical technique involves circumferential lesion dissection, always attempting en-bloc resection to avoid intraoperative bleeding. Our favorable outcomes suggest that microsurgical resection is the treatment of choice for hemangioblastomas of the posterior fossa. Patients with VHL disease should be followed closely for a long-term period, as delayed lesion growth may occur.
- Chen CL, Yu HH (2014) Clinical features and surgical outcomes of sporadic cerebellar hemangioblastomas. Clin Neurol Neurosurg 125: 160-165.
- Amano T, Tokunaga S, Shono T, Mizoguchi M, Matsumoto K, et al. (2009) Cerebellar hemangioblastoma manifesting as hearing disturbance. Neurol Med Chir (Tokyo) 49: 418-420.
- Wan JJ, Hua C, Yong W (2011) Surgical management of large solid hemangioblastomas of the posterior fossa. J Clin Neurosci 18: 39-42.
- Slater A, Moore NR, Huson SM (2003) The natural history of cerebellar hemangioblastomas in von Hippel-Lindau disease. Am J Neuroradiol 24: 1570-1574.
- David W, Andrews (2011) Our current knowledge of hemangioblastomas and treatment. World Neurosurg 75: 45-46.
- Sayer FT, Nguyen J, Starke RM, Yen CP, Sheehan JP (2011) Gamma knife radiosurgery for intracranial hemangioblastomas–outcome at 3 years. World Neurosurg 75: 99-105, 145-108.
- Jian Ch, Wenke L, Si Z, Ding L, Xuhui H (2017) Clinical features and surgical outcomes in patients with cerebellopontine angle hemangioblastomas: Retrospective series of 23 cases. world neurosurg 103: 248-256.
- Marin K, Dragan J, Bruno S, Frederick AB, Kenan IA (2018) Hemangioblastomas of the posterior cranial fossa in adults: demographics, clinical, morphologic, pathologic, surgical features, and outcomes. A systematic review. World Neurosurg 110: e1049-e1062.
- Jagannathan J, Lonser RR, Smith R, DeVroom HL, Oldfield EH (2008) Surgical management of cerebellar hemangioblastomas in patients with von Hippel-Lindau disease. J Neurosurg 108: 210.
- Bush ML, Pritchett C, Packer M, Ray CA, Jacob A (2010) Hemangioblastoma of the cerebellopontine angle. Arch Otolaryngology Head Neck Surg 136: 734-738.
- Hua C, Jian Z, Ying HB, Ming SW, Yong W (2017) Surgical treatment of solid hemangioblastomas of the posterior fossa: A report of 28 cases oncology letters 13: 1125-1130.
- Matsushima T, Kawashima M, Masuoka J, Mineta T, Inoue T (2010) Transcondylar fossa (supracondylar transjugular tubercle) approach: Anatomic basis for the approach, surgical procedures, and surgical experience. Skull Base 20: 83-91.
- Dow GR, Sim DW, O'Sullivan MG (2002) Excision of large solid hemangioblastomas of the cerebellopontine angle by a skull base approach. Br J Neurosurgery 16: 168-171.
- Glasker S, Vortmeyer AO, Lonser RR, Lubensky IA, Okamoto H, et al. (2006) Proteomic analysis of hemangioblastoma cyst fluid. Cancer Biol Ther 5: 549-453.
- Wanebo JE, Lonser RR, Glenn GM, Oldfield EH (2003) The natural history of hemangioblastomas of the central nervous system in patients with von Hippel–Lindau disease. J Neurosurg 98: 82-94.
- Lee GJ, Jung TY, Kim IY, Jung S, Jang WY, et al. (2014) The clinical experience of recurrent central nervous system hemangioblastomas. Clin Neurol Neurosurg 123: 90-95.
- Byung HM, Sang KP, Young MH (2014) Large solid hemangioblastoma in the cerebellopontine angle: complete resection using the transcondylar fossa approach. Brain Tumor Res Treat 2: 128-133.
- Bishop FS, Liu JK, Chin SS, Fults DW (2008) Recurrent cerebellar hemangioblastoma with enhancing tumor in the cyst wall: Case report. Neurosurg 62: E1378-E1389.
- Cornelius JF, Maurice JPS, Bresson D, George B, Houdart E (2007) Hemorrhage after particle embolization of hemangioblastomas: Comparison of outcomes in spinal and cerebellar lesions. J Neurosurg 106: 994-998.
- Eskridge JM, McAuliffe W, Harris B, Kim DK, Scott J, et al. (1996) Preoperative endovascular embolization of craniospinal hemangioblastomas. Am J Neuroradiol 17: 525-531.
- Agarwal V, Babu R, Grier J, Adogwa O, Back A, et al. (2013) Cerebellopontine angle meningiomas: postoperative outcomes in a modern cohort. Neurosurg Focus 35: E10.
- Bamps S, Calenbergh FV, Vleeschouwer SD, Loon JV, Sciot R, et al. (2013) What the neurosurgeon should know about hemangioblastoma, both sporadicand in Von Hippel–Lindau disease: A literature review. Surg Neurol Int 4: 145.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Allergy Research (ISSN:2642-326X)
- Journal of Carcinogenesis and Mutagenesis Research (ISSN: 2643-0541)
- Journal of Nursing and Occupational Health (ISSN: 2640-0845)
- Journal of Cancer Science and Treatment (ISSN:2641-7472)
- Advance Research on Endocrinology and Metabolism (ISSN: 2689-8209)
- Journal of Otolaryngology and Neurotology Research(ISSN:2641-6956)
- Journal of Psychiatry and Psychology Research (ISSN:2640-6136)



