2454
Views & Citations1454
Likes & Shares
Histopathology
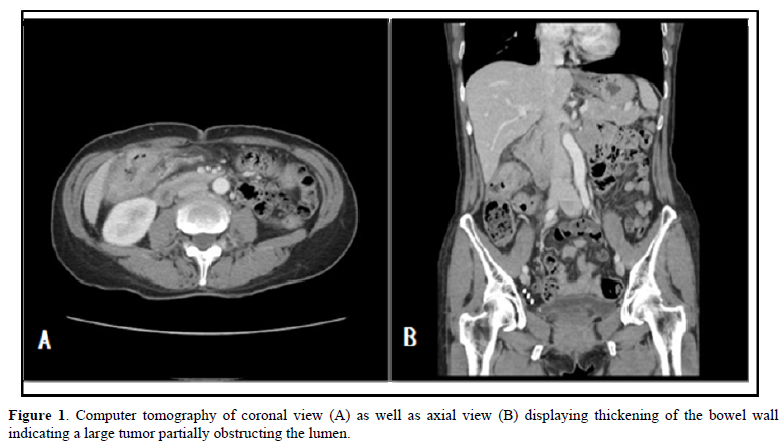
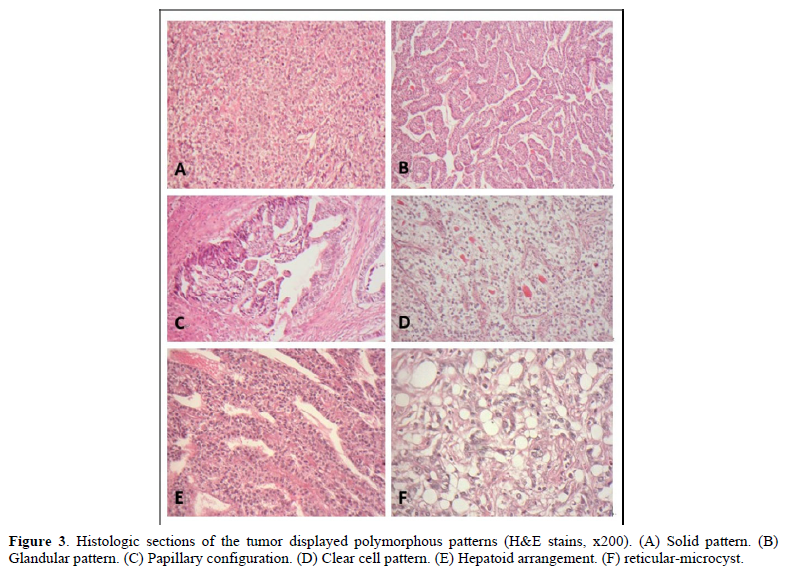
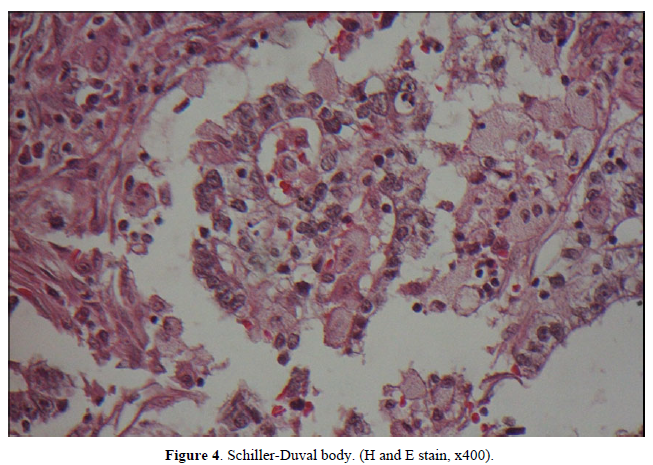
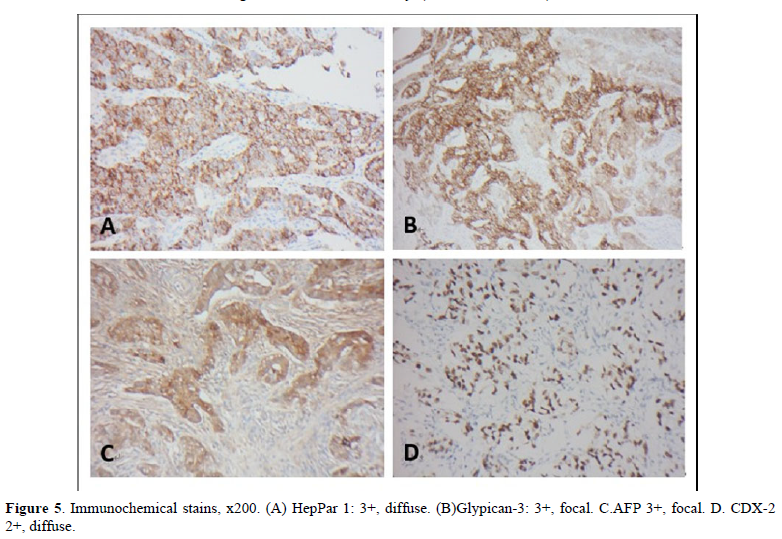
The postoperative specimen consisted of a segment of colon attached with an appendix. On opening, there was a fungating tumor with central ulceration; measuring 4x3x2 cm, four cm from the proximal resection margin (Figure 2). The tumor was grayish white in color, and apparently has invaded through the muscle layer to the serosa. Viewing from the back side, the serosa appeared hemorrhagic with fibrous adhesive bands, indicating adhesion to the adjacent organs. Sections from the tumor displayed a polymorphous appearance, i.e. the tumor was composed of solid sheets with areas of glandular or papillary structure (Figure 3). The solid sheets consist of closely packed cells with eosinophilic or clear cytoplasm. In addition, they exhibited a variety of other patterns characteristic for YST: clear cell, hepatoid and reticular microcystic. A few Schiller-Duval bodies were observed (Figure 4). Immunochemical staining (IHCs) (Figure 5) showed the following results: HepPar 1 3+, diffuse; Glypican-3+, focal; AFP 3+, focal; CDX-3 +, diffuse. HCG was negative (not shown). Histological evaluation, in conjunction with IHC studies, we made this case as yolk sac tumor. Because the tumor invaded through the visceral peritoneum, with six lymph nodes positive out of seventeen examined. There was no radiological evidence of distant metastasis. This tumor was staged as pT4aN2a. Later, we submitted the whole tumor for histological examination, no conventional adenocarcinoma or choriocarcinoma components were found which led us to diagnose this tumor as pure yolk sac tumor of the colon.
DISCUSSION AND LITERATURE REVIEW
AFP-GC accounts for the majority of AFP-producing cancers. AFP-GC is considered as the subtype of gastric cancer, because it has propensity to metastasize to the liver and lymph node, resulting in poor prognosis. AFP-CRC behaves aggressively, similar to that of AFP-GC. The cellular and molecular mechanism responsible for their poor prognosis was not clearly understood. An early report indicated that AFP had a suppressive effect on lymphocyte transformation [8]. Another report indicated that AFP could enhance proliferative activity and increase angiogenesis due to positive expression of vascular endothelial growth factor VEGF [9]. In a study in 2000, Amemiya [10] found that hepatocyte growth factor (HGF) and c-met expression was higher in AFP-GC than that in AFP negative gastric cancer. Through the HGF and c-met pathway, tumor growth was promoted. Patients with AFP-CRC behaved poorly similar to patients with AFP-GC, perhaps through the same mechanism. AFP-CRC is extremely rare, since Nakajima et al, reported the first case of AFP-CRC in 1985 [11], only a few cases have been sporadically published in the literature [12], the largest series was from Ren [13]. They analyzed 20 patients on clinicopathological features and prognosis. None of their cases had yolk sac tumor. According to Otani [14], only four cases of colorectal adenocarcinoma with YST component have been reported in the English literature up to 2022, but they were traditional adenocarcinoma combined with YST, or with YST and choriocarcinoma. To the best of our knowledge, our present case is the first report of pure colonic YST.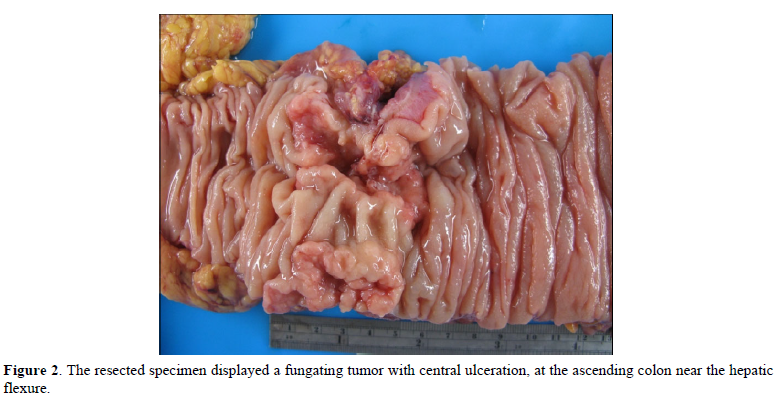
It is worth mentioning that endodermal sinus and yolk sac tumor are used interchangeably. The pathogenesis of the yolk sac tumor in the colon cancer still remains unknown. One theory proposed a retrodifferention mechanism of mature cells into primitive pluripotent cells [15]. A case report by Otani [14] supported this theory. In their patient, the cells from the original adenocarcinoma and yolk sac tumor in the recurrence shared mutation in APC and TP53 genes. The second theory was that of the migration misplacement. During embryogenesis, the primitive gonadal cells migrated from the cranial cavity to the external genitalia. During their migration, some primitive germ cell might misplace at the migration pathway. These cells could transform into germ cell tumor [15]. Currently, there is no standard guideline to treat AFP-CRC patients with germ cell component. In the literature, they were usually treated according to traditional adenocarcinoma, receiving adjuvant chemotherapy after resection. It was promising that some patients could benefit from preoperative chemotherapy [16-20].
CONCLUSION
In conclusion, we reported the first case of pure yolk sac tumor in colon, in which primary tumor resection and lymph dissection were performed. It was unfortunate that our patient was transferred to a medical center for further therapy. We missed the opportunity to gain the knowledge about treating pure yolk sac tumor of the colon.
- Mclntire KR, Waldmann TA, Moertel CG, Go VL (1975) Serum alpha-fetoprotein in patients with neoplasms of the gastrointestinal tract. Cancer Res 35: 991-996.
- Ishukura H, Fukasawa Y, Ogasawara K, Natori T, Tsukada Y, et al. (1985) An AFP- producing gastric carcinoma with features of hepatic differentiation. A case report. Cancer 56: 840-848.
- Khalid Y, Dasu N, Reja, Suga H, Itidiare M, et al. (2020) An extremely rare presentation of an alpha-fetoprotein-producing esophageal adenocarcinoma. Case Rep Gastroenterol 14: 497-503.
- Miyake M, Ito M, Mitsuoka A, Taki T, Wada H, et al. (1987) Alpha-fetoprotein and human chorionic gonadotropin-producing lung cancer. Cancer 59: 227-232.
- Harano K, Ando M, Sasajima Y, Yunokawa M, Yonemori K, et al. (2012) Primary yolk sac tumor of the omentum: A case report and literature review. Case Rep Oncol 5: 671-675.
- Li XD, Wu CP, Ji M, Wu J, Lu B, et al. (2013) Characteristic analysis of a-fetoprotein- producing gastric cancer in China. World J Surg Oncol 11: 246e51.
- Takashi Y, Mai O, Orgino T, Ueda H, Sawaguchi K, et al. (1987) Clinicopathological study of AFP producing gastric cancer. Jpn Surg Soc 88: 696-700.
- Yachim S (1983) the immunosuppressive properties of alpha-fetoprotein. A brief review. NY Acad Sci 417: 105-107.
- Kamei S, Kono K, Amemiaya H, Takahashi A, Sugai H, et al. (2003) Evaluation of VEGF and VEGF-C expression in gastric cancer cells producing a-fetoprotein. J Gastroenterol 38: 540-547.
- Amemiya H, Kono K, Mori Y, Takahashi A, Ichihara F, et al. (2000) High frequency of c-Met expression in gastric cancer producing alpha-fetoprotein. Oncology 59: 145-151.
- Nakajima T, Okazaki T, Morinaga S, Tsumuraya M, Shimosato Y, et al. (1985) A case of alpha-fetoprotein-producing rectal carcinoma. Jpn J Clin Oncol 15: 679-685.
- Karakuch N, Toyota K, Kajikawa R, Wanatabe A, Saito Y, et al. (2021) Alpha-fetoprotein-producing colon carcinoma with rapidly increasing liver metastasis that resulted in death: A case report. J Anus Rectum Colon 5: 100-106.
- Ren F, Weng W, Zhang Q, Tan C, Xu M, et al. (2019) Clinicopathological features and prognosis of AFP-producing colorectal cancer: A single center analysis of 20 cases. Cancer Manag Res 11: 4557.
- Otani T, Kanemura H, Kimura M, Matsmura N, Sato T, et al. (2022) Yolk Sac tumor in a recurrence of colonic adenocarcinoma with shared mutation in APC and TP53 Genesis: A case report. Int J Surg Pathol 30: 646-651.
- Nogales FP, Preda O. Nicolae A (2012) Yolk sac tumor revisited. A review of their many faces and names. Histopathology 60: 1023-1033.
- Nakamura Y, Matsuda K, Yokoyama S, Tamura K, Mitani Y, et al. (2018) Alpha-fetoprotein producing rectal cancer successfully respond to preoperative chemotherapy. Surg Case Rep 4: 11.
- Yu YY, Ogino T, Okada S (1992) an alpha-fetoprotein-producing carcinoma of rectum. Acta Pathol Jpn 42: 684-687.
- Kawahara M, Takada A, Tachibana A, Kodama T, Kobayashi H, et al. (2009) Germ cell tumor of the colon with an adenomatous component. Int J Clin Oncol 14: 537-540.
- Ostor AG, McNaughton WM, Fortune DW, Rischin D, Hill coat BL, et al. (1993) Rectal Carcinoma with germ cell elements treated with chemotherapy. Pathology 25: 243-246.
- Petricek CM (2001) Colonic adenocarcinoma: Metastasizing as a germ cell neoplasm. A Case report and review of the literature. Arch Pathol Lab Med 125: 558-561.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- Journal of Alcoholism Clinical Research
- Ophthalmology Clinics and Research (ISSN:2638-115X)
- Oncology Clinics and Research (ISSN: 2643-055X)
- Journal of Immunology Research and Therapy (ISSN:2472-727X)
- Journal of Cell Signaling & Damage-Associated Molecular Patterns
- Journal of Clinical Trials and Research (ISSN:2637-7373)
- Journal of Cardiology and Diagnostics Research (ISSN:2639-4634)






