2813
Views & Citations1813
Likes & Shares
CASE REPORT 2: ID H10441, 1965
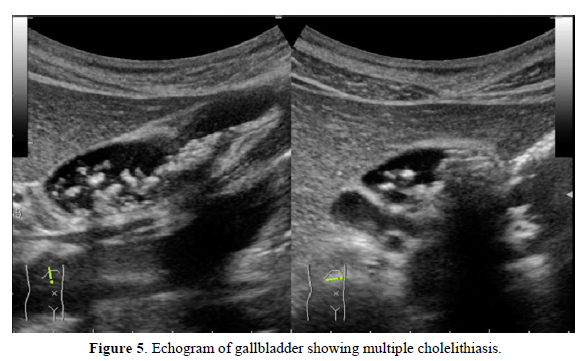
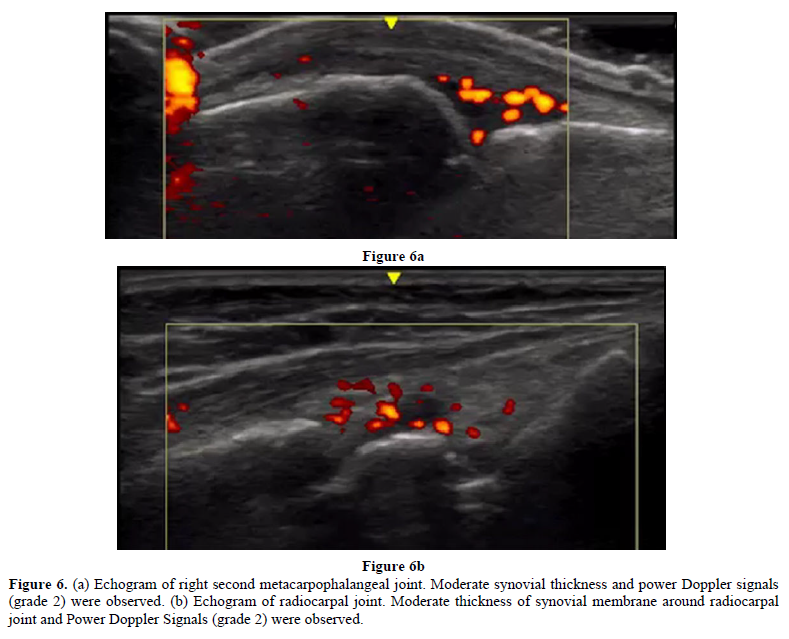
In March 2016, a female patient visited the clinic complaining of palpitations, fatigue, and amenorrhea. Low-dose conjugated equine estrogen-medroxyprogesterone acetate was prescribed (Figure 4). In December 2016 and December 2017, she experienced colicky right upper abdominal pain. An abdominal ultrasound showed cholelithiasis, so she stopped receiving HRT to undergo a cholecystectomy. At the end of December 2017, she complained of abrupt severe joint pain in the knees and wrists and difficulty performing activities of daily living. She had not undergone the cholecystectomy. In January 2018, the patient resumed HRT. Her joint pains gradually decreased and her quality of life improved. She visited the orthopedic clinic for further evaluation of her joint pain. The patient was prescribed methotrexate for RA based on a high titer of anti-CCP Ab (128.4/mL), but she did not take the medication. In April 2018, she presented to our clinic. Neither joint swelling nor C-reactive protein elevation were present, so cyclic HRT (E2 patch and dydrogesterone) was recommenced to prevent RA. Her joint pain disappeared completely. In April 2019, a slight elevation of C-reactive protein and multiple small gallstones were observed (Figure 5). The patient underwent cholecystectomy and did not receive HRT for two weeks. In May 2019, she experienced peripheral joint pain and swelling, and a joint ultrasound showed a power Doppler signal of grade 2 (Figures 6a & 6b). Methotrexate (8 mg/week) was added to her HRT regime. She has remained well.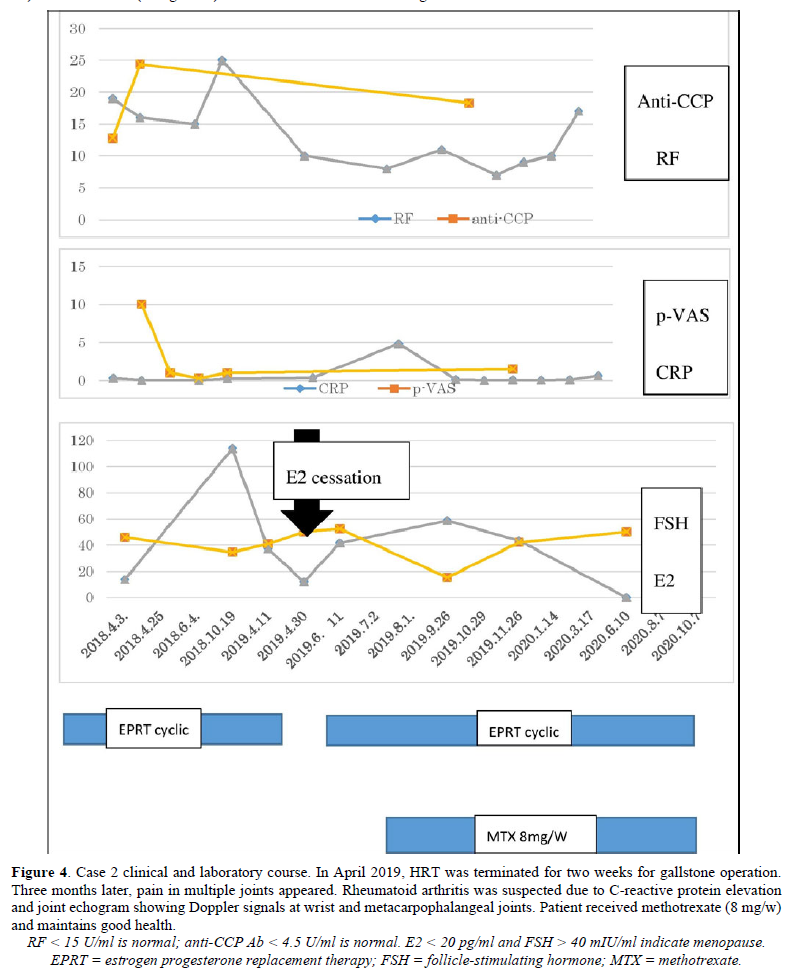
DISCUSSION
HRT use is not common in Japan, in part due to the 2002 Women’s Health Initiative report highlighting its adverse effects. In general, HRT is recommended in younger, healthy women for no longer than 5 years [8]. If the benefits of HRT outweigh its risks, HRT can continue for more than 5 years in women younger than 60 years. The duration and termination of HRT depends on the patients’ overall health and individual factors [9].
Over the last 20 years, menopausal women with morning stiffness, arthralgia, or positive ANA have used HRT for relief of joint pain and to prevent RA or other autoimmune rheumatic diseases [10,11]. SS demonstrates a female preponderance (male: female ratio 1:14-1:17), similar to primary biliary cirrhosis [12]. Like RA, SS onset frequently occurs between the ages of 40 to 60, when estrogen levels decrease. Childbirth appears to be a protective factor, with parous women age
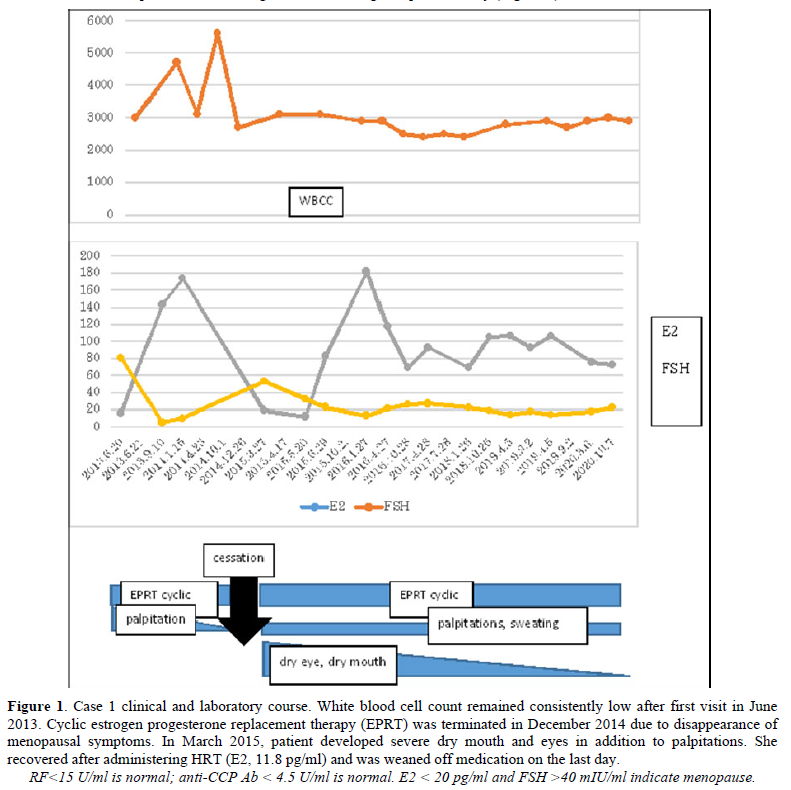
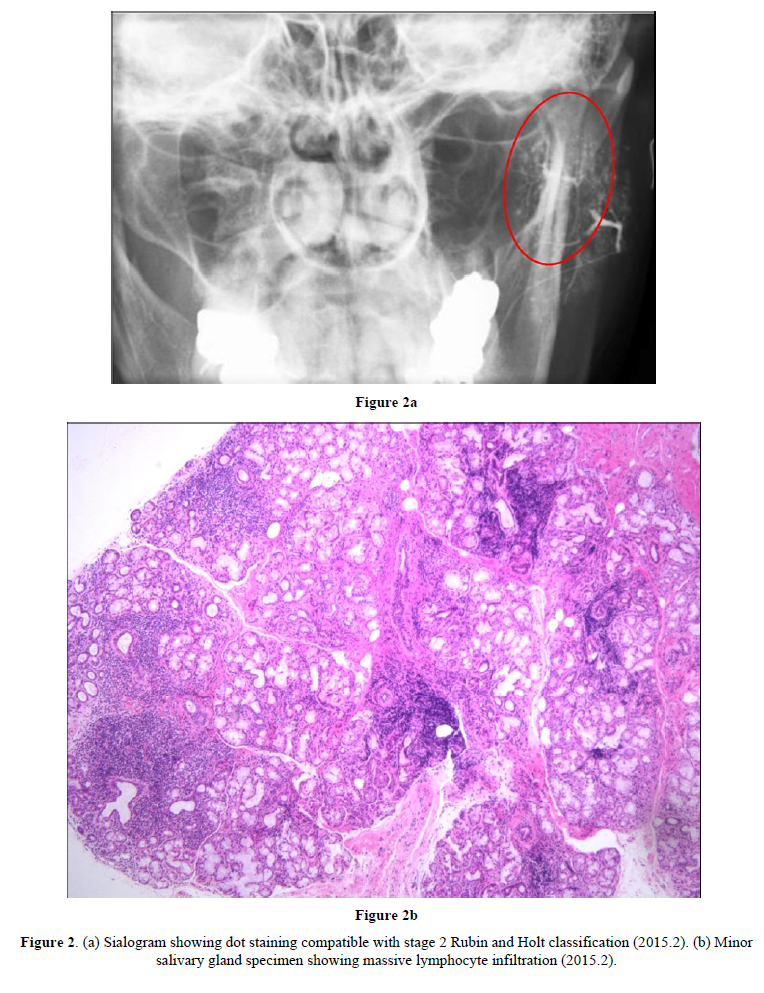
The cause of SS is not clearly understood. Genetic background likely plays a role, but in one study of identical twins, the odds of developing SS in both were only 15% [16]. Other causes could be environmental, particularly infection [17], or individual-level factors. In case 1, after termination of HRT, ANA was positive at a 1:1280 dilution and anti-SS-A was positive at a 1:64 dilution according to micro-Ouchterlony, suggesting escalation of autoantibody production due to estrogen deficiency [18]. Although sialography and minor salivary biopsy were not tested prior to symptom development, it is possible that cessation of HRT induced clinical SS directly.
Recently, the reverse specific sequence oligopeptide [19] method has identified HLADRB1 *04:05 and *15:02. The significance of HLADRB1 15 [20] and HLADRB1 *15:02 in SS remains unknown, however. Further research is needed to determine the role, if any, of HLADRBI 15. In case 1, HRT was a viable treatment option for preventing RA because of HLADRB1*04:05 positivity.
Estrogen deficiency or depletion is linked with peri-and post-menopausal symptoms, as well as autoimmune disease and osteoporosis [21] Pacifici R et al recently reported alteration of gut microbe induce osteoporosis [22]. Walitt et al. reported that HRT was not useful for preventing RA; however, the results may have been affected by high BMIs, smoking status, and age (average 63.5 years old) of participants at commencement of HRT [23]. In 1947, the average age of menopause onset in Japan was 50 years. As people live longer, the risk of autoimmune diseases will likely increase more for women than for men. Estrogen deficiency or depleted activation of NFkB via inhibition of kB-Ras2 is regulated via miR-125b and let-7a, which produce TNF [5] and activate Th 17 cells, [24] which can lead to the development of RA.
Our reports here are limited to two cases with symptomatic SS or RA. Further studies are needed to assess E2 and FSH fluctuation before and after onset of these conditions. In conclusion, abrupt cessation of HRT for 1 to 3 months led to onset of SS and RA in two cases, respectively. It is possible that HRT may prevent progression to RA and SS from undifferentiated arthralgia.
FUNDING
None
CONFLICT OF INTEREST
None
ACKNOWLEDGMENTS
We thank Dr. Nakagawa and Dr. Saito at the Tsurumi Dental University for the evaluation of Sjogren’s syndrome.
ORCID identifier: 0000-0002-9143-3738, 0000-0002-2019-9040
- Writing Group for the Women's Health Initiative Investigators, et al. (2002) Risks and benefits of estrogen plus progestin in healthy postmenopausal women: Principal results from the Women's Health Initiative randomized controlled trial. JAMA 288(3): 321-333.
- Ness J, Aronow WS, Beck G (2006) Menopausal symptoms after cessation of hormone replacement therapy. Maturitas 53(3): 356-361.
- Lindh-Astrand L, Bixo M, Hirschberg AL, Sandstrom-Poromaa I, Hammar M (2010) A randomized controlled study of taper-down or abrupt discontinuation of hormone therapy in women treated for vasomotor symptom. Menopause 17(1): 72-79.
- Feitsma AL, van der Voort EIH, Franken KLC, Bannoudi H, Elferink BG, et al. (2010) Identification of citrullinated vimentin peptides as T cell epitopes in HLA-DR4-positive patients with rheumatoid arthritis. Arthritis Rheum 62(1): 117-125.
- Murphy AJ, Guvre PM, Pioli PA (2010) Estradiol suppresses NF kappa B activation through coordinated regulation of Let-7 and miR-125b in primary human macrophages. J Immunol 184: 5029-5037.
- Melby MK (2006) Climacteric symptoms among Japanese women and men: Comparison of four symptom checklists. Climacteric 9(4): 298-304.
- London: National Institute for Health and Care Excellence (UK). National Institute for Health and Clinical Excellence: Guidance.
- Cagnacci A, Venier M (2019) The controversial history of hormone replacement therapy. Medicina (Kaunas) 55(9): 602.
- Crawford, SL, Crandall CJ, Derby CA, Khoudary SRE, Waetjen LE, et al. (2018) Menopausal hormone therapy trends before versus after 2002: Impact of the Women's Health Initiative Study Results. Menopause 26(6): 588-597.
- Miyachi K, Sasse B, Ihara A (2018) Does hormone replacement therapy prevent undifferentiated arthritis progressing to rheumatoid arthritis? Ann Rheum Dis pp: 540-541.
- Miyachi K, Sasse B, Nomoto S, Igarashi T, Mashiba S, et al. (2019) The treatment of women with postmenopausal undifferentiated arthralgia- the first report of efficacy of hormone replacement therapy. Am J Biomed Sci Res 1: 124-129.
- Patel R, Shahane A (2014) The epidemiology of Sjogren’s syndrome. Clin Epidemiol 6: 247-255.
- Guthrie KA, Dugowson CE, Voigt LF, Koepsell TD, Nelson JL (2010) Does pregnancy provide vaccine-like protection against rheumatoid arthritis? Arthritis Rheum 62(7): 1842-1848.
- Ito M, Kojima T, Miyata M, Saka M, Kokubun M, et al. (1995) Primary biliary cirrhosis (PBC)-CREST (calcinosis, Raynaud's phenomenon, esophageal dysfunction, sclerodactyly and telangiectasia) overlap syndrome complicated by Sjögren's syndrome and arthritis. Intern Med 34(5): 451-454.
- Mattey DL, González-Gay MA, Hajeer AH, Dababneh A, Thomson W, et al. (2000) Association between HLA-DRB1*15 and secondary Sjögren's syndrome in patients with rheumatoid arthritis. J Rheumatol 27(11): 2611-2616.
- Anaya JM, Delgado-Vega AM, Castiblanco J (2006) Genetic basis of Sjögren's syndrome. How strong is the evidence? Clin Dev Immunol 13(2-4): 209-222.
- Hitchon CA, Chandad F, Ferucci ED, Willemze A, Ioan-Facsinay A, et al. (2010) Antibodies to porphyromonas gingivalis are associated with anticitrullinated protein antibodies in patients with rheumatoid arthritis and their relatives. J Rheumatol 37(6): 1105-1112.
- Miyachi K, Naito M, Maeno Y, Suzuki S, Hamamoto T, et al. (1983) Sialographic study in patients with and without antibodies Sjogren’s syndrome A(Ro). J Rheumatol 10: 387-394.
- Ayo CM, da Silveira Camargo AV, Xavier DH, Batista MF, Carneiro OA, et al. (2015) Frequencies of allele groups HLA-A, HLA-B and HLA-DRB1 in a population from the northwestern region of São Paulo State, Brazil. Int J Immunogenet 42(1): 19-25.
- Kaushansky N, Eisenstein M, Boura-Halfon S, Hansen BE, Nielsen CH, et al. (2015) Role of a novel human leukocyte antigen-DQA1*01:02;DRB1*15:01 mixed isotype heterodimer in the pathogenesis of ‘humanized’ multiple sclerosis-like disease. J Biol Chem 290(24): 15260-15278.
- Pacifici R, Browns C, Puscheck E, Friedrich E, Slatopolsky E, et al. (1991) Effect of surgical menopause and estrogen replacement on cytokine release from human blood mononuclear cells. Proc Natl Acad Sci 88: 5134-5138.
- Yi LJ, Yu M, Pal S, Tyagi AM, Dar H, et al. (2020) Parathyroid hormone-dependent bone formation requires butyrate production by intestinal microbiota. J Clin Invest 130(4): 1767-
- Walitt B, Pettinger M, Weinstein A, Katz J, Torner J, et al. (2008) Effects of postmenopausal hormone therapy on rheumatoid arthritis: The Women’s Health Initiative randomized controlled trials. Arthritis Care Res 59: 302-310.
- Andersson A, Stubelius A, Karlsson MN, Engdahl C, Erlandsson M, et al. (2015) Estrogen regulates T helper 17 phenotype and localization in experimental autoimmune arthritis. Arthritis Res Ther 17(1): 32.
QUICK LINKS
- SUBMIT MANUSCRIPT
- RECOMMEND THE JOURNAL
-
SUBSCRIBE FOR ALERTS
RELATED JOURNALS
- International Journal of Radiography Imaging & Radiation Therapy (ISSN:2642-0392)
- Journal of Blood Transfusions and Diseases (ISSN:2641-4023)
- Journal of Otolaryngology and Neurotology Research(ISSN:2641-6956)
- International Journal of Diabetes (ISSN: 2644-3031)
- Advance Research on Alzheimers and Parkinsons Disease
- Archive of Obstetrics Gynecology and Reproductive Medicine (ISSN:2640-2297)
- International Journal of Internal Medicine and Geriatrics (ISSN: 2689-7687)







